Welcome to EM Cases’ CritCases blog, a collaboration between Mike Betzner, the STARS air ambulance service and EM Cases’ Michael Misch and Anton Helman! These are educational cases with multiple decision points where there is no strong evidence to guide us. Various strategies and opinions from providers around the world are coalesced and presented to you in an engaging format. Enjoy!
Written by Michael Misch, edited by Anton Helman, March 2016
The Case:
A 44 year-old male presents to Janus General, a small town rural emergency department with the chief complain of feeling unwell for 3 weeks. He endorses general malaise, cough, shortness of breath and chest pressure. In addition, he complains of chills, myalgias and loose stools during this time. He denies any travel or sick contacts.
Past medical history is remarkable for hypertension for which he is no longer taking medication. He has a 20 pack-year history of smoking cigarettes and regularly uses crack cocaine and methamphetamine. He also admits to heavy alcohol use. He denies IV drug use or history of HIV or Hepatitis.
On exam, he appears quite anxious and has increased work of breathing. Vitals are: BP 100/43, HR 140-170, RR 36-40, , temperature 36.5, oxygen saturation 95% room air. Heart sounds are normal. JVP is flat. There is reduced air entry bilaterally but the lungs are clear. Abdomen is soft, ENT exam unremarkable. No signs of IV drug use.
Initial Investigations are obtained:
Initial blood work including venous blood gas
| Na | 125 | pH | 7.16 |
| K | 4.3 | pCO2 | 42 |
| Cl | 90 | HCO3 | 15 |
| HCO3 | 15 | pO2 | 20 |
| Glc | 10.6 | Lactate | 8.8 |
| Base Excess | – 13 | ||
Initial CXR
POCUS shows a hyperdynamic heart with a normal right ventricle and no pericardial effusion. IVC suggests hypovolemia. There are no B-lines on lung POCUS.
What is your differential diagnosis?
There is a huge differential in this patient. One useful framework is based on the type of shock:
- Septic Shock: Even though this patient is afebrile, sepsis should certainly be a consideration. With the respiratory symptoms, pneumonia would be a likely source. CXR suggests possible bilateral infiltrates.
- Obstructive Shock: Massive pulmonary embolism should be considered, however one would expect the patient to be more hypoxic if the PE is large enough to account for this degree of tachycardia.
- Cardiogenic Shock: The patient has cardiac risk factors for ACS. Clinically the patient did not appear volume overloaded as his JVP was flat. His CXR also does not suggest pulmonary edema. Compensated cardiogenic shock + wide complex tachycardia is a possibility.
Specific Differential:
1) Myocarditis – Consistent with the insidious onset, the low grade chills and overall SOB and chest pain findings; would account for the tachycardia (although I would expect him to be hypotensive to get a lactate of 9).
2) Septic Shock with pneumonia – Common things are common. Dehydrated and little to see initially on the CXR but may reveal itself with fluid resuscitation. Would account for the lactate and the
hyponatremia due to SIADH.
3) Pancreatitis – Chronic pancreatitis could certainly give him weight loss and malabsorption
diarrheal stools. Pain and local complications could account for many of the findings, however soft abdominal exam makes this unlikely.
4) PE – unlikely with that oxygen saturation and that severe a tachycardia
5) ACS – I think unlikely given the duration of the symptoms >1 week
6) Pulmonary hypertension from crack use – should have an elevated JVP and RV enlargement on POCUS
7) Boerhaaves – No history of vomiting just nausea
8) Cocaine cardiomyopathy
9) Myasthenia gravis new onset with muscle weakness (respiratory included) triggered by drugs or infection-Dr. Marc N. Francis MD, FRCPC
My question is whether this guy has somehow gotten into some metformin and this is a metformin lactic acidosis?
-Rob Abernethy MD FRCPC
Toxic alcohol ingestion and thyroid storm are other considerations in the differential.
What are your immediate interventions?
The presumed diagnosis based on the initial investigations is sepsis with a respiratory source. The patient is pan-cultured, started on Levofloxacin 750 mg and Vancomycin 1g IV. He receives 4 L of crystalloid before the JVP becomes visible.
1) IV bolus 250cc NS with low threshold to give more if tolerating same and assess for any change to the rate – would be careful NOT to hammer fluids into this guy.
2) I personally would not cardiovert this rhythm at this time. I am unconvinced that it is VT and the patient has reasonable vital signs in spite of the tachycardia. If he is a myocarditis there is the possibility of this degrading further with cardioversion. I am not saying I would not consider cardioversion at some point, but I would likely not go there right out of the gate.
3) If my fluids did nothing to the rate and I was certain it was not sinus tachycardia than I would try gentle IV B-blockers (low dose 1-2mg metoprolol or IV esmolol).
4) Broad spectrum antibiotics – Ceftriaxone and Vancomycin
5) Echogardiogram -Marc N. Francis MD, FRCPC
Consider steroids (query Addisonian crisis given hypovolemia-like state and hypoNa) if not responding well to initial measures. – Chris Hall MD FRCPc
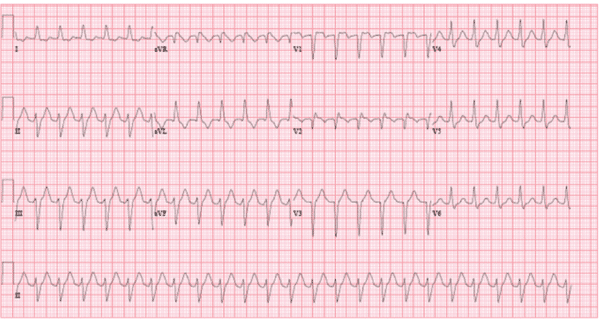
What is your interpretation of the above EKG? What steps will you take to further elicit the rhythm?
This patient is in a monomorphic wide complex tachycardia with a rate of 145. The QRS is 126 ms. There are no obvious p waves or flutter waves on the ECG.
Differential: Sinus tachycardia, AVNRT, atrial flutter with aberrancy or ventricular tachycardia. Variation in the rate would make SVT less likely.
I’m gonna have to go with atrial flutter as the most likely rhythm. I think I see flutter waves in lead l. I wouldn’t bet the farm on it, however, and as this patient is quite sick, cardioversion would not be a bad way to go (even though it would be James (Jamie) Fox, MD, FRCPC
The initial rhythm on the monitor does not look that wide to me. Even on the later EKG it is not “super-wide”. It certainly could be a sinus tachycardia with aberrancy at the rates seen. Could also be SVT and I would entertain atrial flutter with a 2:1 block + aberrancy. -Rob Abernethy MD FRCPC
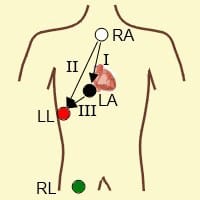
Lewis Leads: Differentiating Atrial from Ventricular Tachydysrhythmias
While not done in this case, the use of Lewis Leads may help to bring out atrial activity and help to determine the underlying rhythm. To use this technique:
- Place the right arm lead to the right of the sternum at the 2nd intercostal space.
- Place the left arm lead to the right of the sternum at the 4th intercostal space
- Inspect the ECG for atrial activity in lead l
Consider the use of Lewis Leads when you have a monomorphic wide complex tachycardia and atrial activity is not clearly demonstrated on a standard 12-lead ECG.
For a more in-depth explanation of the use of Lewis leads to differentiate SVT with aberancy from VT see this great FOAMed Paramedic blog: My Variables Only Have 6 Letters
What is your interpretation of the venous blood gas?
The VBG is interesting – aside from the obvious lactate and the acidosis it appears that he is not appropriately compensating from a respiratory point of view to his metabolic acidosis. The rate of 36-40 seems adequate but the PCO2 is normal? Superimposed respiratory acidosis? Opiates on board? -Rob Abernethy MD FRCPC
Use adenosine for regular monomorphic rhythms only
The use of adenosine may be considered in regular monomorphic rhythms in an attempt to slow AV conduction and bring out the atrial activity. The use of adenosine should only be considered in regular monomorphic rhythms. Patients presenting in atrial fibrillation with an accessory pathway will present in an irregularly irregular wide complex rhythm. The refractory period of the AV node prevents ventricular rates in excess of 150-170 beats per minute. The use of adenosine in this scenario will block conduction down the AV node and encourage conduction down the accessory pathway, which can precipitate conduction at excessive rates into the ventricle and potentially cause ventricular fibrillation. While adenosine has been traditionally taught to only convert atrial rhythms such as SVT, there is a well-described subset of electrophysiologically-proven ventricular tachycardia that will cardiovert with adenosine; so called “adenosine-responsive ventricular tachycardia”. Thus, conversion to normal sinus rhythm using adenosine should not be used as a diagnostic tool to differentiate SVT with aberancy from Vtach.
Case Continued…
Clinically, this man’s respiratory distress does not improve following initial interventions. His heart rate is climbing to persistently above 150. Air transport is requested.
Vitals post 4.5 L crystalloid T 36.9, HR 160, BP 104/55, RR 40 SaO2 95% 4L NP
Further investigations at this point are resulted:
| Na | 122 | Hb | 141 | Billi | 16 | Acetaminophen | < 66 |
| K | 4.4 | Lk | 31.1 | ALP | 102 | Ethanol | 12 |
| Cl | 86 | Plt | 133 | LDH | 546 | Salicylates | < 0.07 |
| Cr | 148 | GGT | 54 | ||||
| Glc | 10.3 | Lipase | 17 | ||||
| Trop | 627 ng/L | ||||||
| Urea | 12.5 | ||||||
Repeat VBG 90 Mins later:
| Na | 125 | pH | 7.25 |
| K | 4.4 | pCO2 | 28 |
| Cl | 90 | HCO3 | 12 |
| HCO3 | 15 | pO2 | 29 |
| Glc | 10.1 | Lactate | 7.1 |
| Base Excess | – 14 |
Repeat CXR
Based on the investigations above, what is your differential diagnosis now? What are your next steps for this patient?
The very high white count might further support infectious cause. The markedly elevated troponin should prompt consideration for myocarditis, especially in context of significant tachycardia. However, POCUS showed grossly normal systolic function. PE should still be considered but one would expect hypoxia with the degree of tachycardia and hypotension caused by the clinical presentation. The repeat CXR more strongly suggests bilateral infiltrates, which in a drug-abuser should prompt consideration of Pneumocystis Jerovicii pneumonia (especially with the elevated LDH). Finally, these infiltrates could represent septic emboli from an endocardititis. As per Dr Carr’s Best Case ever on endocarditis, if endocarditis is considered, vancomycin and ceftriaxone should be given following at least 3 sets of blood cultures.
Case Continued…
The patient’s hemodynamics fail to improve following a significant fluid challenge. The rhythm is not clear at this time. While en route to the tertiary care centre, the heart rate rises to 160-170 with a BP of 90/40.
Would you electrically cardiovert this patient with the information you have at this point?
Case Continued…
Given the significant tachycardia and hemodynamic instability without a clear underlying cause, synchronized cardioversion is attempted. The patient is given boluses of 10-20 mg of propofol in rapid succession until sedation is achieved. Synchronized cardioversion with 100 J, 150J, and then 200 J is attempted but is not successful, making the diagnosis of sinus tachycardia more likely. He is subsequently started on a norepinephrine infusion. Upon arrival at the tertiary centre, cardiology and ICU are consulted. Cardiology is similarly unable to definitely determine the presenting rhythm although given failed cardioversion, it is presumed to be sinus tachycardia.
What is the underlying diagnosis in this patient in septic shock?
Case Continued…
This 44 year-old man is admitted to the ICU with piperacillin-tazobactam added to the antimicrobial regimen. CT chest confirms the presence of bilateral lung infiltrates. Streptococcus Pneumoniae grows in his blood after 6 hours. An echocardiogram reveals vegetations on a bicuspid aortic valve. Additionally, there is severe aortic regurgitation as well as an aortoatrial fistula tracking from an area beyond the aortic valve back to the left atrium. The final diagnosis is endocarditis with probable pneumonia complicated by an aortoatrial fistula.
How does endocarditis with acute aortic regurgitation account for this presentation?
This man presented in mixed septic and cardiogenic shock from endocarditis complicated by acute aortic regurgitation (AR).
In patients with native aortic valves, acute AR is due to endocarditis or aortic dissection. With chronic AR, the left ventricle can dilate to accommodate for regurgitant flow, which maintains end diastolic pressure and cardiac output. In acute AR, however, the left ventricle is not able to dilate to accommodate for increased end diastolic pressure, which decreases stroke volume and cardiac output. Coronary ischemia results from decreased coronary blood flow during diastole, and is exacerbated by increased myocardial oxygen demand. The increased myocardial oxygen demand results from tachycardia and increased end diastolic pressures against which the left ventricle must contract. This would account for the markedly elevated troponin in this case.
His severe ongoing tachycardia was a result of very poor forward flow. His heart was beating the heck out of itself trying to get any forward flow at all. -M.J. Betzner MD FRCPc
Due to the pathophysiologic difference of acute and chronic AR, patients with acute AR may not present with classic signs of a holosystolic murmur and a decreased pulse pressure (which are typical of chronic AR). Instead, there is often only a soft decrescendo murmur, which may be easily missed when the patient is tachycardic and tachypneic. Pulse pressure is normal or may be decreased. Patients will present with dyspnea, hemodynamic instability and shock.
The presentation in this particular case is complicated by septic shock. S. Pneumoniae is an uncommon cause of endocarditis, accounting for 1-3% of cases. However it is particularly aggressive with a mortality rate approaching 25%. It is thought to occur most commonly in middle aged-men with a significant alcohol intake (as in this case). Two thirds of cases occur in patients without previous valvular pathology. S. Pneumoniae causes severe valve damage with one study finding a 20% rate of valve perforation and 13% rate of peri-valve abscess. In this same cohort, two thirds of patients required valve replacement. Rarely, S. Pneumoniae can cause the triad of endocarditis, meningitis and pneumonia known as Austrian (or Osler’s) Triad.
What is the definitive management for this patient with endocarditis and acute aortic regurgitation?
The definitive management of acute aortic regurgitation is surgical valve replacement. Medical management to bridge the gap to urgent surgery is two-fold:
1. Decrease afterload with a nitroprusside infusion
2. Increase contractility and cardiac output with dobutamine
While an intra-aortic balloon pump is sometimes used in cardiogenic shock, its use is contraindicated in AR because balloon inflation during diastole worsens regurgitant flow.
Case Resolution…
This man has acute endocarditis, of a bicuspid aortic valve, secondary to S. Pneumoniae as well as a probable pneumonia. Consistent with S. Pneumoniae endocarditis, there is significant valvular damage requiring emergent valve replacement.
Take Home Points
- Consider the use of Lewis Leads to accentuate atrial activity on ECG when faced with a tachydysrythmia of unknown origin.
- The use of adenosine should only be considered in regular monomorphic rhythms. A trial of adenosine with a regular monomorphic wide complex tachycardia may help to elicit p waves to different VT from SVT with abberancy. However, cardioversion with adenosine should not be used to diagnose SVT with aberrancy because a subset of electrophysiologically-proven VT will cardiovert with adenosine.
- Acute AR may present as cardiogenic shock. The classic murmur and pulse pressure of chronic AR may be absent. These patients require urgent valve replacement or repair.
- S. Pneumoniae is a relatively rare but devastating cause of endocarditis, which requires aggressive pneumococcal antibiotic coverage and often, urgent valve replacement.
For David Carr’s Top Ten Pearls and Pitfalls on Endocarditis see his Best Case Ever
Dr. Helman, Dr. Misch and Dr. Betzner have no conflicts of interest to declare
References
Bakker AL, Nijkerk G, Groenemeijer BE, et al. The Lewis lead: making recognition of P waves easy during wide QRS complex tachycardia. Circulation. 2009;119(24):e592-3. Full Text
Kanakadandi V, Annapureddy N, Agarwal SK, et al. The Austrian syndrome: a case report and review of the literature. Infection. 2013;41(3):695-700.
Lefort A, Mainardi JL, Selton-suty C, Casassus P, Guillevin L, Lortholary O. Streptococcus pneumoniae endocarditis in adults. A multicenter study in France in the era of penicillin resistance (1991-1998). The Pneumococcal Endocarditis Study Group. Medicine (Baltimore). 2000;79(5):327-37.
Wilber DJ, Baerman J, Olshansky B, Kall J, Kopp D. Adenosine-sensitive ventricular tachycardia. Clinical characteristics and response to catheter ablation. Circulation. 1993;87(1):126-34. Abstract
Additional FOAMed Resources
Salim Rezaie reviews differentiating SVT with aberrancy vs VT on R.E.B.E.L. EM
David Carr reviews management of endocarditis on EMCrit






Easy to say in retrospect however AR significant enough to contribute to his clinical presentation should be obvious with color flow doppler at his initial echo.
To my knowledge, aortic reurgitation is associated with widened pulse pressure, not decreased. please correct me if I am wrong.