This is Part 1 of EM Cases two part podcast on TIA and Stroke with Walter Himmel and David Dushenski – TIA Update – Risk Stratification, Workup and Dual Antiplatelet Therapy.
Much has changed in recent years when it comes to TIA risk stratification, workup and antiplatelet therapy. In this podcast we use the overarching theme of timing to elucidate how to distinguish true TIA from the common TIA mimics, the importance of timing in the workup of TIA, why the duration of therapy with dual antiplatelet therapy and timing of starting anticoagulation in patient with atrial fibrillation, contributes to the difference between preventing catastrophic strokes and causing intracranial hemorrhage. Remember that stroke is a leading cause of adult disability and is the third leading cause of death in Canada. It’s time we paid more attention to TIA…
Podcast production, sound design & editing by Anton Helman
Written Summary and blog post by Shaun Mehta & Alex Hart, edited by Anton Helman November, 2018
Cite this podcast as: Helman, A, Himmel, W, Dushenski, D. TIA Update – Risk Stratification, Workup and Dual Antiplatelet Therapy. Emergency Medicine Cases. November, 2018. https://emergencymedicinecases.com/tia-update/. Accessed [date]
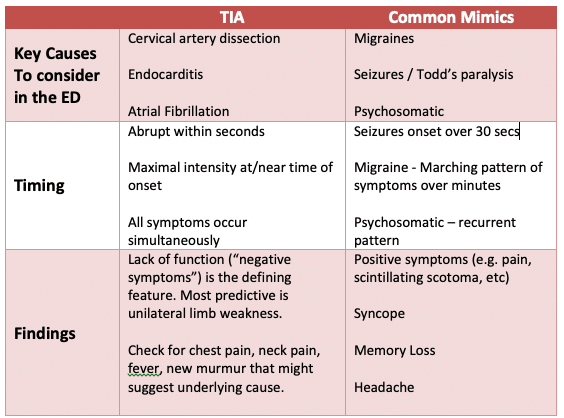
TIA Mimics
The ED misdiagnosis rate of TIA has been reported to be between 5% and 31%, which can lead to needless workups, iatrogenic complications and morbidity. The three mimics that are often not obvious on history and physical are migraines, seizures and psychiatric disorders. Distinguishing these from TIA is mostly in the timing of symptom onset, associated symptoms and presence of “positive” vs “negative” symptoms. It is important to consider a few of the less common underlying causes of TIA in the ED as these have time-sensitive workups and therapies.
TIA Important Causes and Common Mimics
An observational study from 2014 shows that memory loss, headache, and blurred vision were associated with mimics, while unilateral weakness was associated with true TIA.
Clinical Pearl: The “TIA AND” presentation. TIA symptoms AND neck pain – think neck dissection. TIA symptoms AND fever or new heart murmur – think endocarditis.
TIA risk stratification – the death of the ABCD2 score
The importance of risk stratification for TIA lies in the questions: what’s the chance that a TIA patient you see in the ED will have a stroke in the next 2 days? 90 days? And can we identify the patients who are eligible for a carotid endarterectomy fast enough to prevent that stroke?
There exists an alarming 12-20% 90 day stroke risk in those presenting with high risk TIA symptoms. Half of these patient suffer from a stroke in the first 48hrs. A 2016 NEJM study fortunately found that this high risk can be reduced to less than 4% with rapid follow-up and aggressive secondary prevention. Based on these findings, it is good practice for high risk TIA patients to be worked up in the first 48 hours.
So which patients who present with TIA symptoms are high risk?
While the CHADS2VASC helps identify AFib patients at risk for future embolic event, The ABCD2 score has been the most ubiquitously used risk stratifying tool in ED since its inception. The elements of the ABCD2 include:
- Age over 60
- Initial BP over 140/90
- Clinical features of unilateral weakness and speech impairment
- Duration of symptoms
- History of D
More recent studies by Stead and Ghia have failed to validate the ABCD2 score and have shown that the score is neither sensitive nor specific and is inaccurate, at any cut-point. In 2011, Perry’s external validation of the ABCD2 found that by using this score, physicians were misclassifying up to 8% of patients as low risk. Sensitivity of the score for high risk patients was found to be only 31.6%.
As recognized in the latest iteration of the Canadian Heart and Stroke Guideline from 2018, the most important prognostic feature of the ABCD2 score appears to be the Clinical features:
“Very High Risk for Recurrent Stroke (Symptom onset within last 48 Hours):
- Transient, fluctuating or persistent unilateral weakness (face, arm and/or leg);
- Transient, fluctuating or persistent language/speech disturbance;
- And/or fluctuating or persistent symptoms without motor weakness or language/speech disturbance (e.g. hemibody sensory symptoms, monocular vision loss, hemifield vision loss, +/- other symptoms suggestive of posterior circulation stroke such as binocular diplopia, dysarthria, dysphagia, ataxia).”
The bottom line: high risk patients are the ones with either true motor deficit of a limb or face or a speech deficit. This is important not only to determine who needs a rapid workup to assess eligibility for carotid endarterectomy and hopefully prevent a catastrophic stroke, but also to identify those patients who will likely benefit from dual antiplatelet therapy.
Update 2021: The Canadian TIA score was validated using a multicenter prospective cohort study involving 7607 patients in the ED with TIA or minor stroke, and was found to more accurately stratify the risk of subsequent stroke and carotid endarterectomy / stenting when compared to the ABCD2 and ABCD2i scores. Abstract
Workup of high risk TIAs – the time to investigate is now, with CTA
CT angiogram of the head and neck improves identification of clinically significant stenosis amenable to endarterectomy compared to carotid doppler ultrasound as well as diagnose both carotid and vertebral artery dissection as a cause for the TIA.
Based on Canadian Stroke Best Practices Guidelines 2018
High risk patients: “Urgent brain imaging (CT or MRI) and non-invasive vascular imaging (CT angiography (CTA) or MR angiography (MRA) from aortic arch to vertex should be completed as soon as possible within 24 hours (Evidence Level B)”
Moderate risk patients*: a) CT/CTA or MRI/MRA (aortic arch to vertex), b) If you don’t have quick access to CTA, an ultrasound of the carotids is an acceptable option.
*Moderate risk = “Fluctuating or persistent symptoms without motor weakness or language/speech disturbance (e.g., hemibody sensory symptoms, monocular vision loss, binocular diplopia, hemifield vision loss, dysarthria, dysphagia, and / or ataxia).”
Which TIA patients need an urgent echocardiogram and/or holter monitor?
Cardio-embolic pathology are a non-trivial source of TIAs and stroke. Five percent of patients with TIA will have atrial fibrillation found on cardiac monitor within 24hrs. 15-20% will have paroxysmal atrial fibrillation on cardiac monitoring within 4 weeks. It is important for these patients to be identified early so that appropriate anticoagulation therapy can be started to prevent further cardio-embolic phenomena.
Those patients being admitted will likely have a full work-up as an inpatient. For patients being considered for outpatient management, which of them requires a holter monitor and echocardiogram to help guide management?
Two groups of TIA patients require urgent echocardiogram and holter monitor:
1. Patients with known heart disease including rheumatic heart disease, heart failure, severe valvular disease, severe CAD or history of MI.
2. Patients with no obvious cause of their TIA and no classic risk factors to identify an underlying cause of their TIA such as paroxysmal atrial fibrillation, severe valvular disease including endocarditis, PFO etc.
Dual antiplatelet therapy (DAPT) for TIA – again, timing is everything
Platelet antagonism is the cornerstone of TIA therapy. Knowing what to give and, more importantly, when to give it has not always been so clear. Timing your therapy can make the difference between preventing an ischemic event and causing a life-threatening hemorrhage.
MATCH Trial
The MATCH trial in 2004 studied DAPT compared to clopidogrel alone in 7600 patients. This study found little benefit for DAPT and did note a clinically significant increase in major bleeds. However, the inclusion criteria were for patients having had a TIA in the preceding 6 months. The treatment period was for up to 2 years.
EXPRESS Trial
The EXPRESS trial in 2007 clearly demonstrated that early management of TIA results in 80% reduction in early stroke risk. This early therapy was not a priority in the MATCH trial.
FASTER, CHANCE, POINT Trials
Three more recent trials helped demonstrate that the major benefit of DAPT exists early in the course of treatment and the major risks occur later. If DAPT was started appropriately early (usually within 24-72hrs) and not extended beyond 3 weeks, a 1.5-3.5% decreased risk of stroke was found, without significant increased risk of major bleeding.
Our experts therefore recommend immediate loading of DAPT (unless already taking ASA or clopidogrel) for high risk patients and dosing in the following fashion:
Load with ASA 160-325mg chewed followed by ASA 81mg po daily
Load with Clopidogrel 300mg po followed by 75mg po daily for 3 weeks only
Clinical Pearl: For those patients receiving 3 weeks of dual antiplatelet therapy, prescribe a PPI for gastric mucosal protection, our experts recommend pantoprazole because the other PPI’s affect the cytochrome P450 system and may impair the activation of clopidogrel.
Timing of anticoagulation for patients who you know have atrial fibrillation when they present with a TIA
There’s that timing theme again. If you’re sure it’s a TIA with full recovery within one hour (no residual symptoms) and a normal CT, consider starting anticoagulation (DOAC or warfarin) in the ED. If you’re not sure, consult your neurologist.
It is important to understand that for patients with a completed ischemic stroke, with known atrial fibrillation, who are not anticoagulated at the time of their stroke, anticoagulation is not indicated immediately in the ED. Rather, an anticoagulant should be started at a later time during their admission depending on the severity of the stroke.
Disposition for TIA – Which patients should be admitted and which patients can have outpatient management?
We don’t think twice about admitting stroke patients who present outside the thrombolytic window. However, it is high-risk TIA patients that may benefit most from admission for a rapid workup and endarterectomy that may prevent a catastrophic stroke. We can save a high-risk TIA patient from having a massive CVA, but once they’ve stroked out, the benefit of admitting them is far from huge.
Practically, if you can get a CTA in the ED, and it’s negative, outpatient management within 48hrs is reasonable.
If CTA shows carotid stenosis amenable to endarterectomy, admission is advisable. Time is essential – the highest risk time is the first two days and then the first two weeks. Therefore, surgery should be done with 2-14 days after the TIA – the sooner the better. This reduces the risk of a major stroke from 26% over 2 years to 9% – a 17 % absolute risk reduction. The absolute risk reduction is 30% if done within 2 weeks.
Learn more about an updated approach to stroke in the ED in Episode 120: ED Stroke Management in the Age of Endovascular Therapy
References
- Boulanger JM, Lindsay MP, Gubitz G, et al. Canadian Stroke Best Practice Recommendations for Acute Stroke Management: Prehospital, Emergency Department, and Acute Inpatient Stroke Care, 6th Edition, Update 2018. Int J Stroke. 2018;:1747493018786616. Full pdf
- Nadarajan V, Perry RJ, Johnson J, Werring DJ. Transient ischaemic attacks: mimics and chameleons. Pract Neurol. 2014;14(1):23-31.
- Amarenco P, Lavallée PC, Labreuche J, et al. One-Year Risk of Stroke after Transient Ischemic Attack or Minor Stroke. N Engl J Med. 2016;374(16):1533-42.
- Valls J, Peiro-chamarro M, Cambray S, Molina-seguin J, Benabdelhak I, Purroy F. A Current Estimation of the Early Risk of Stroke after Transient Ischemic Attack: A Systematic Review and Meta-Analysis of Recent Intervention Studies. Cerebrovasc Dis. 2017;43(1-2):90-98.
- Perry JJ, Sharma M. Prospective validation of the ABCD2 score for patients in the emergency department with transient ischemic attack. CMAJ. 2011 Jul 12;183(10):1137-45.
- Stead LG, Suravaram S. An assessment of the incremental value of the ABCD2 score in the emergency department evaluation of transient ischemic attack. Ann Emerg Med. 2011 Jan;57(1):46-51.
- Schrock JW, Victor A, Losey T. Can the ABCD2 risk score predict positive diagnostic testing for emergency department patients admitted for transient ischemic attack? Stroke. 2009 Oct;40(10):3202-5.
- Ghia D, Thomas P. Low positive predictive value of the ABCD2 score in emergency department transient ischaemic attack diagnoses: the South Western Sydney transient ischaemic attack study. Intern Med J. 2012 Aug;42(8):913-8.
- Rothwell PM, Giles, MF, Chandratheva A, Marquardt L, Geraghty O, Redgrave JNE et al.Effect of urgent treatment of transient ischaemic attack and minor stroke on early recurrent stroke (EXPRESS study): a prospective population-based sequential comparison. Lancet. 2007;370:1432-1442.
- Diener HC, Bogousslavsky J, Brass LM, Cimminiello C, Csiba L, Kaste M, Leys D, Matias-Guiu J, Rupprecht HJ. Aspirin and clopidogrel compared with clopidogrel alone after recent ischaemic stroke or transient ischaemic attack in high-risk patients (MATCH): randomised, double-blind, placebo-controlled trial. Lancet. 2004;364(9431):331-7.
- Kennedy J, Hill MD, Ryckborst KJ, et al. Fast assessment of stroke and transient ischaemic attack to prevent early recurrence (FASTER): a randomised controlled pilot trial. Lancet Neurol. 2007;6(11):961-9.
- Johnston SC, Easton JD, Farrant M, Barsan W, Conwit RA, Elm JJ, Kim AS, Lindblad AS, Palesch YY. Clopidogrel and Aspirin in Acute Ischemic Stroke and High-Risk TIA. N Engl J Med. 2018 Jul 19;379(3):215-225.
- Wang Y, et al. Clopidogrel with aspirin in acute minor stroke or transient ischemic attack. N Engl J Med. 2013 Jul 4;369(1):11-9.
- Kapral MK, Hall R, Fang J, et al. Association between hospitalization and care after transient ischemic attack or minor stroke. Neurology. 2016;86(17):1582-9.
Other FOAMed Resources on TIA
EM Docs on risk stratification
EM Literature of Note on disposition of TIA
Drs. Helman, Himmel and Dushenski have no conflicts of interest to declare
Now test your knowledge with a quiz.





Thanks so much for that episode! Really looking forward to changing parts of my practice. Wondering if you would be willing to share the order set for TIA/CVA that you guys use at North York General?
Thanks so much much for this podcast! Certainly practice changing for me. Wondering if you would be able to share your order set for North York General for stroke?
THANKS FOR TEH FRUIUTFUL TALKS , I HAVE ONE QUESTION : I got a pregnant lady with the 9th month with TIA loss of speech for about 45 min ……is imaging safe at this point and what the benefit and risks of the dual antiplatelet therapy?
Imaging is a shared decision making process. There is a radiation risk to the fetus but it is very tiny. DAPT has not been studied in pregnant patients to my knowledge. I’d consult a neurologist.
Please, would you clarify what to when a patient presents with what sounds like a TIA, normal head CT and already on either Aspirin or Clopidogrel? The centre I work in (UK) is not using DAPT. Do you recommend giving a loading dose of the one not being used and then continuing it at maintenance dose and stopping the other?