This is EM Cases Episode 120 – ED Stroke Management in the Age of Endovascular Therapy
According to the Global Burden of Disease Study published in NEJM in December 2018 the estimated lifetime risk for a 25 year old during their remaining lifespan is 25% [1]. Stroke is the 3rd leading cause of death and 1st leading cause of major disability in North America. As we transition from the relatively simple era of systemic thrombolytics under 3 hours vs “ASA and admit” for over 3 hours, decision making has become much more complicated and varied, depending on where you work. Which patients need what type of scanning? Where should that scanning be done – at the stroke center or at the peripheral center? Which patients should get systemic thrombolytics? Which patients should get endovascular therapy? In this EM Cases main episode podcast, a follow up to our episode on TIA released in November 2018 with Walter Himmel and David Dushenski, we try to simplify the confusing time-based and brain tissue-based options for stroke management…
Podcast production, sound design & editing by Anton Helman
Written Summary and blog post by Anton Helman, January 2019
Cite this podcast as: Helman, A, Himmel, W, Dushenski, D. ED Stroke Management in the Age of Endovascular Therapy. Emergency Medicine Cases. January, 2019. https://emergencymedicinecases.com/stroke-update-endovascular-therapy/. Accessed [date]
Time and image based stroke management algorithm
Activating a “code stroke” on every patient that experiences any acute neurologic event within 24hrs of symptom onset based on the DIFFUSE 3 and DAWN trials [2,3] may outstrip resources, with only a tiny minority of these patients receiving potential benefit. There is currently an effort to identify those patients clinically who might be most likely to benefit from endovascular therapy so that not all stroke patients require transport to a stroke center with multiple imaging modalities and resource-heavy acute stroke team care.
The workup and considerations for tPA and endovascular therapy (thrombectomy) depend on:
- Symptom onset to needle time
- Type of stroke, NIHSS or VAN tool (see below)
- CT, CT angiogram and CT perfusion results
- Contraindications to tPA/thrombectomy
Time is the key factor in patients with a symptom onset-to-needle time ≤ 6 hours.
Brain tissue salvageability determined by CT perfusion is the key factor in patients with a symptom onset-to-needle time of 6-24 hours.
Acute Stroke Management Time & Tissue Based Algorithm
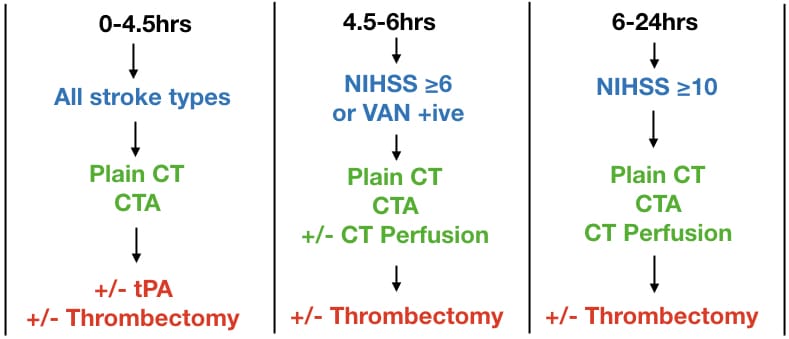
Times are symptom onset to needle times at a stroke center. CTA = CT angiogram head and neck. NIHSS = National Institute of Health Stroke Scale. VAN = Vision, Aphasia, Neglect tool. NIHSS on MD Calc
In the 0-4.5 hours category, plain CT will rule in hemorrhagic stroke and CTA may diagnose an underlying vascular abnormality causing the bleed.
For patients in the 4.5-6 hours category, and with an NIHSS ≥6 or VAN positive, and plain CT and CTA consistent with cortical stroke, candidacy for thrombectomy is determined either by CT perfusion, or if CT perfusion is not readily available, by the ASPECTS score[6].
Update 2021: Retrospective cohort study across 1752 USA hospitals of 163,038 patients with acute ischemic stroke, either taking DOAC’s or not taking anticoagulants prior to stroke and treated with IV alteplase within 4.5 hours of symptom onset. Use of DOAC’s within the preceding 7 days, compared to no use of anticoagulants, was not significantly associated with increased risk of intracranial hemorrhage. Abstract
Update 2022: A systematic review including 7 trials from 2012 to 2020 with 980 patients with “wake-up” strokes identified that 46% of patients who underwent endovascular thrombectomy had independent functional outcome (modified Rankin scale of 2 or less) compared to 9% of patients who were randomized to control (RR 5.12, 95% CI 2.57 to 10.17, P<0.001). The review also found that 66% of patients who underwent IV alteplase had functional outcome (modified Rankin scale of 2 or less) compared to 58% randomized to control (RR 1.13, 95% CI 1.01 to 1.26, P = 0.03). Abstract
Using the VAN Tool to identify strokes that may be amenable to endovascular therapy
Which patients are most likely to benefit from endovascular therapy? Stroke patients with large vessel occlusions (“cortical stroke”) are the most likely to benefit from thrombectomy – internal carotid artery, MCA, and some ACA occlusions. An NIHSS ≥6 has traditionally been used to predict a cortical stroke. However this score is complicated and time consuming for the ED provider and ideally requires specific training to use it. Multiple prehospital prediction tools have been developed such as the LAMS and RACE , however their sensitivities for large vessel strokes were only in the 80’s. A more recent tool called the VAN tool (a mnemonic for Vision, Aphasia and Neglect) has been shown in a pilot study to have a 100% sensitivity and 90% specificity, 74% PPV and 100% NPV for large vessel stroke [7]. VAN was more specific than an NIHSS score ≥6 and just as sensitive for large vessel occlusion stroke. Although this study requires validation it is a promising tool to help us in the ED to identify clinically which patients should be considered for advanced imaging and endovascular therapy.
VAN Tool to identify large vessel cortical strokes
Patient must have weakness plus one or all of the V, A, or N to be VAN positive.
Step 1: Weakness
Weakness – ask the patient to raise both arms up for 10 seconds and assess for drift, weakness or paralysis; if any of these are present proceed to step 2
Step 2: V or A or N
Visual disturbance – field cut, diplopia or blindness
Aphasia – expressive (repeat and name 2 objects) or receptive (unable to follow commands – close eyes or make a fist)
Neglect – inability to track to one side, ignoring one side, unable to feel both sides at the same time or unable to identify own arm
Medications for stroke patients who do not fulfill criteria for tPA or endovascular therapy
There is no role for dual antiplatelet therapy in completed stroke, unless the NIHSS ≤3 and CT or MRI show a small area of hypodensity < 1-2 cm diameter (very minor stroke). Starting with the CAST trial in 1997 and based on two more recent meta-analyses in 2012 and 2013, the risk of hemorrhagic transformation outweighs any potential benefit in most patients with completed stroke [8,9,10].
There is no role for starting anticoagulant medications in the ED for patients with history of atrial fibrillation or who present with atrial fibrillation at the time of their stroke. Again, the risk of hemorrhagic transformation outweighs any potential benefit of starting anticoagulants in the acute phase.
The risk of recurrent stroke in patients with atrial fibrillation is approximately 2% per week for the first two weeks after a stroke [4].
At what time after a stroke should patients with atrial fibrillation be started on anticoagulants? Expert opinion consensus suggests that for patients with atrial fibrillation and:
- Large size stroke, anticoagulation should be considered starting on day 10.
- Medium size stroke, anticoagulation should be considered starting on day 5-7.
- Small size stroke, anticoagulation should be considered starting on day 3-5.
Informed consent and shared decision making for thrombolytics and endovascular therapy for stroke
There is a range of opinions as to whether or not informed consent is required for thrombolytics and/or endovascular therapy for stroke. On the one extreme, some stroke neurologists believe that informed consent is not required because they consider these treatments “standard of care”. On the other extreme there are some EM physicians who believe that thrombolytics for stroke have no evidence for benefit and don’t offer these treatments at all. Our experts recommend obtaining informed consent from the patient and/or POA whenever possible.
With regards to endovascular therapy it is important to explain that only approximately 1-2% of patients will qualify and benefit clinically from endovascular therapy.
With regards to thrombolytic therapy it is important to discuss the values of the patient (see Episode 47 EBM with Walter Himmel); whether or not they value life at all costs and whether they consider a possible small improvement worth the risk of getting much worse or dyeing. Explain to the patient and/or POA that some doctors believe that there is a 1 in 6 to 1 in 8 chance that there will be some improvement but on the other hand a 1 in 16 chance that a catastrophic head bleed will occur.
The CAEP stroke position statement recommends discussing and offering tPA for patients who you anticipate a symptom onset to needle time of ≤ 3 hours. For patients between 3 and 4.5 hours, tPA should be offered for patients who present at a stroke center [11].
“Thrombolytic therapy for acute ischemic stroke patients should not be routinely offered for the treatment of acute ischemic stroke for patients if administered beyond three hours of stroke symptom onset (WEAK RECOMMENDATION, MODERATE QUALITY EVIDENCE). The administration of thrombolytic therapy for acute ischemic stroke beyond 3 hours from stroke symptom onset should be restricted to specialized stroke centers with advanced imaging capabilities or as part of a research protocol (WEAK RECOMMENDATION, LOW QUALITY EVIDENCE).”
The ACEP clinical policy recommends discussing and offering tPA for all patients who qualify ≤ 4.5 hours regardless of the center of treatment [12].
2018 Canadian Stroke Guideline Summary
Update 2021: Open-label, multi-center, randomized trial in Europe of stroke patients who received endovascular treatment (EVT) alone or IV alteplase followed by EVT (standard of care in Europe). Across the 539 patients in the study (using the modified Rankin scale, from 0 [no disability] to 6 [death]), neither superiority nor non-inferiority of EVT alone was discovered. Abstract
References
- The GBD 2016 Lifetime Risk of Stroke Collaborators. Global, regional, and country-specific lifetime risks of stroke, 1990 and 2016. N Engl J Med 2018 Dec 20; 379:2429.
- Albers GW, Marks MP, Kemp S, et al. Thrombectomy for Stroke at 6 to 16 Hours with Selection by Perfusion Imaging. N Engl J Med. 2018;378(8):708-718.
- Nogueira RG, Jadhav AP, Haussen DC, et al. Thrombectomy 6 to 24 Hours after Stroke with a Mismatch between Deficit and Infarct. N Engl J Med.2018; 378:11-21.
- Boulanger JM, Lindsay MP, Gubitz G, et al. Canadian Stroke Best Practice Recommendations for Acute Stroke Management: Prehospital, Emergency Department, and Acute Inpatient Stroke Care, 6th Edition, Update 2018. Int J Stroke. 2018;:1747493018786616.
- Powers WJ, Rabinstein AA, Ackerson T, et al. 2018 Guidelines for the Early Management of Patients With Acute Ischemic Stroke: A Guideline for Healthcare Professionals From the American Heart Association/American Stroke Association. Stroke. 2018.
- Barber PA, Demchuk AM, Zhang J, Buchan AM. Validity and reliability of a quantitative computed tomography score in predicting outcome of hyperacute stroke before thrombolytic therapy. ASPECTS Study Group. Alberta Stroke Programme Early CT Score. Lancet. 2000;355(9216):1670-4.
- Teleb MS et al. Stroke Vision, Aphasia, Neglect (VAN) Assessment – A Novel Emergent Large Vessel Occlusion Screening Tool: Pilot Study and Comparison with Current Clinical Severity Indices. J Neurointerv Surg 2017.
- CAST: randomised placebo-controlled trial of early aspirin use in 20,000 patients with acute ischaemic stroke. CAST (Chinese Acute Stroke Trial) Collaborative Group. Lancet. 1997;349(9066):1641-9.
-
Geeganage CM, Diener HC, Algra A, Chen C, Topol EJ, Dengler R, et al. Dual or mono antiplatelet therapy for patients with acute ischemic stroke or transient ischemic attack: systematic review and meta-analysis of randomized controlled trials. Stroke. 2012;43:1058–1066.
-
Wong KS, Wang Y, Leng X, Mao C, Tang J, Bath PM, et al. Early dual versus mono antiplatelet therapy for acute non-cardioembolic ischemic stroke or transient ischemic attack: an updated systematic review and meta-analysis. Circulation. 2013;128:1656–1666.
- Harris D, Hall C, Lobay K, et al. Canadian Association of Emergency Physicians position statement on acute ischemic stroke. CJEM. 2015;17(2):217-26.
- Brown MD, Burton JH, Nazarian DJ, Promes SB. Clinical Policy: Use of Intravenous Tissue Plasminogen Activator for the Management of Acute Ischemic Stroke in the Emergency Department’.Annals of Emergency Medicine 66 (2015) 322-333.e31.
Drs. Helman, Dushenski and Himmel have no conflicts of interest to declare.
Other FOAMed Resources on Stroke Management and Endovascular Therapy
Journal Jam on “Endovascular Therapy for Stroke” (Part 1, Part 2)
First10EM on Interventional Therapy for Acute Ischemic Stroke – The Evidence
The Bottom Line on DAWN and DIFFUSE-3
REBEL EM on Endovascular Therapy for Acute Ischemic Stroke
Now test your knowledge with a quiz.





Great stuff and fantastic work on everything you do.
So is the jury still out on whether a pt receives periprocedural ASA if endovascular thrombectomy is the goal, without tPA involved? So if after negative for bleed on CT/CTA, but the lesion is likely a good candidate for EVT…are your experts giving ASA immediately if the patient is outside the window for tPA?
Maybe it doesn’t even matter and I’m thinking too much into it, since you’ll do this in concert with a center that can perform endovascular therapy and discuss it with the neurologist. Would you wait until a center says they won’t pursue endovascular therapy to give ASA? Guidelines suggest we can let the inpatient team deliver aspirin (within 24-48 hours), unless the patient comes a day after the stroke:
https://www.acc.org/latest-in-cardiology/ten-points-to-remember/2018/01/29/12/45/2018-guidelines-for-the-early-management-of-stroke
The only information I could find is that there is a MR CLEAN-MED for IV ASA:
https://mrclean-med.nl/study-design.html
And this study suggesting better outcomes for ASA pretreatment without increased bleed risk…https://www.ncbi.nlm.nih.gov/pubmed/29259123