This is EM Cases Aortic Dissection Live from The EM Cases Course
While missing aortic dissection was considered “the standard” in the late 20th century, our understanding of the clinical diagnosis has improved considerably since the landmark International Registry of Aortic Dissection (IRAD) study in 2000. Nonetheless, aortic dissection remains difficult to diagnosis with 1 in 6 being missed at the initial ED visit. Why? The diagnosis is rare with and incidence of only 2.9/100,000/year, and the presentation is often atypical mimicking other more common diagnoses such as ACS and stroke.
Each hour that passes from the onset of symptoms portends a 1-2% increase in mortality so your early, timely diagnosis is key. The most important factor leading to a correct diagnosis is having a high clinical suspicion. We need to at least consider the diagnosis in all patients with chest, abdominal or back pain, syncope or stroke symptoms, yet we shouldn’t be working up every one of them, or else we’ll bankrupt the health care system with all the CT aortograms ordered. Herein lies the difficulty.
With the help of David Carr and Anton Helman we’ll discuss how to pick up atypical presentations without over-imaging as well as manage them like pros by reviewing:
1. The five pain pearls
2. The concepts of CP +1 and 1+ CP
3. Physical exam pearls
4. Initial tests pearls and pitfalls
5. The importance of the correct order and aggressive use of IV medications
So with these objectives in mind…
Written Summary and blog post written by Anton Helman, February 2016
This podcast was recorded live at The EM Cases Course
at North York General Hospital in Toronto February 2017
Cite this podcast as: Helman, A, Carr, D. Aortic Dissection Live from The EM Cases Course. Emergency Medicine Cases. February, 2017. https://emergencymedicinecases.com/aortic-dissection-em-cases-course/. Accessed [date].
For core knowledge on aortic dissection first review EM Cases Episode 28 with David Carr and Anil Chopra
A) The Five Pain Pearls of Aortic Dissection
Pain Pearl #1: Ask the following 3 questions for all patients with torso pain:
- Quality of pain (most commonly “sharp” but highest LR for “tearing”)
- Pain intensity at onset
- Radiation of pain (back and/or belly)
A 1998 study that reviewed a series of aortic dissection cases showed that for the 42% of physicians who asked these 3 questions, the diagnosis was suspected in 91%. When less than 3 questions were asked dissection was suspected in only 49%.
Pain Pearl #2: Think of aortic dissection as the subarachnoid hemorrhage of the torso

Pain Pearl #3: Severe colicky chest pain + opioids = heightened suspicion
If you find yourself treating your chest pain patient with IV opioids to control their severe colicky pain, think about the possibility of aortic dissection.
Pain Pearl #4: Migrating pain has a +LR = 7.6
In addition to the old adage “pain above and below the diaphragm should heighten your suspicion for aortic dissection”, severe pain that progresses and moves in the same vector as the aorta, increases the likelihood of aortic dissection.
Pain Pearl #5: The pain can be intermittent as dissection of the aortic intima stops and starts
The combination of severe migrating and intermittent pain should raise the suspicion for aortic dissection.
Painless Aortic Dissection
While IRAD reported a painless aortic dissection rate of about 5%, a more recent study out of Japan reported that 17% of aortic dissection patients had no pain. These patients presented more frequently with a persistent disturbance of consciousness, syncope or a focal neurologic deficit. Cardiac tamponade was more frequent in the painless group as well.
B) The Concept of CP +1 and 1+ CP in Aortic Dissection
Big red devascularizes
The intimal tear in the aorta can devascularize any organ from head to toe including the brain, heart and kidneys, and so 5% of dissections present as strokes, and these certainly are not the kind of stroke patients who should be receiving tPA! An objective focal neurologic deficit in the setting of acute unexplained chest pain has +LR = 33 for aortic dissection. Some of the CP +1 phenomena to think about include:
CP + CVA
CP + paralysis
CP + hoarseness (recurrent laryngeal nerve)
CP + limb ischemia
In addition to thinking of CP +1 it may help to think backwards in time and ask the patient who presents with end organ damage if they had torso pain prior to their end organ damage symptoms. For example, for patients who present with stroke symptoms, ask them if they had chest pain, back pain or abdominal pain before the stroke symptoms.
C) Physical Exam Pearls in Aortic Dissection: Look, Listen & Feel
Anyone under the age of 40 years who presents to the ED with unexplained torso pain should be asked if they have Marphan’s Syndrome. In the IRAD under 40 years of age subgroup analysis, 50% of the aortic dissection patients had Marfan’s representing 5% of all dissections.

- Arachnodactyly (Elongated fingers): if they look long try to elicit the wrist sign (see this 15 second video here)
- Pectus excavatum : sternal excavation (see image)
- Lanky limbs
Listen. A new aortic regurgitation murmur has a surprisingly high +LR = 5.
Feel. Feel for a pulse deficit which has a +LR = 2.7.
BP Pitfall: Do not assume that the patient with a normal or low BP does not have an aortic dissection. We know from the IRAD data that only about half of patients are hypertensive at initial presentation. Dissection that progress into the pericardium and end up with tamponade are often hypotensive.
BP Pearl: Patients with dissection who have a wide pulse pressure should be considered pre-terminal, and usually require immediate surgery.
What about bilateral BP differences in aortic dissection?
It’s important to know that 19 % of the general population have a BP difference between arms > 20 and 53 % > 10. This is important to take into account when coming up with a pretest probability.
D) Initial Tests: CXR, Troponin, D-dimer & POCUS
It’s not only about wide mediastinum in CXR interpretation of aortic dissection
1/3 of CXRs in aortic dissection are normal to untrained eye, and a common pitfall is to assume that if the CXR is normal that the patient does not have an aortic dissection.
Tips on reading CXR for aortic dissection:
1st compare to an old CXR to see if there’s been an interval change.
2nd look for more than just a wide mediastinum.
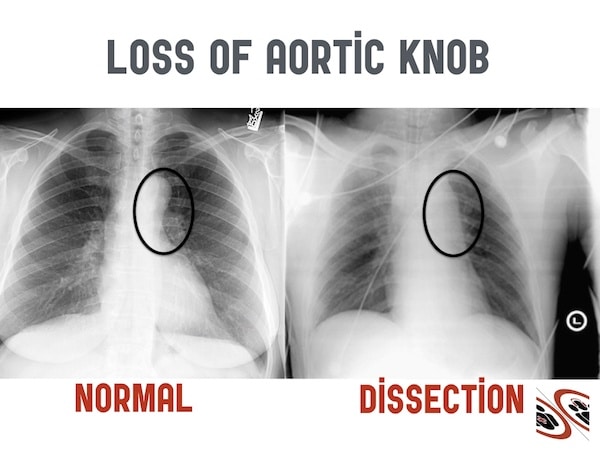
There are about a dozen x-ray findings, but two of them are especially important: Loss of the aortic knob/aortic-pulmonary window and the calcium sign.
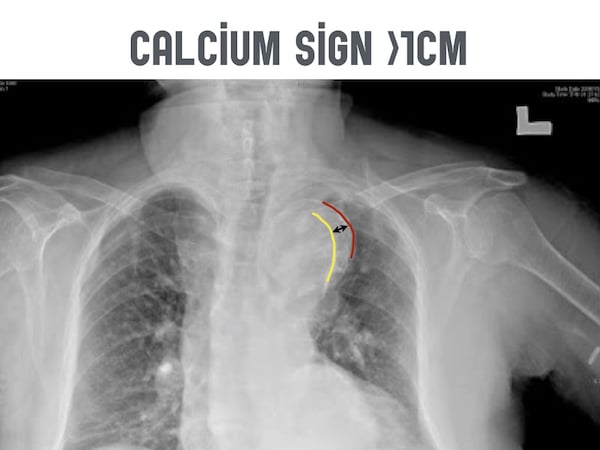
Look for a white line of calcium within the aortic knob. Then measure the distance from there to the outer edge of the aortic knob.
A distance >0.5cm is considered a positive calcium sign and a distance >1.0cm is considered highly suspicious for aortic dissection.
Blood Test interpretation for Aortic Dissection
What are the most common pitfalls in the assessment of aortic dissection when it comes to blood test interpretation?
Troponin
About 1/4 of Type A dissections will have a positive troponin, so if you suspect the diagnosis based on other clinical findings, don’t assume isolated ACS when the troponin comes back positive. Remember that < 1/100 of of patients with a dissection will have associated coronary ischemia in any coronary distribution (most commonly inferior). Another pitfall is working up a disproportionate number of your STEMI patients for aortic dissection causing delays to STEMI treatment. Workup for aortic dissection in STEMI patients should be reserved for those patients with a significant pretest probability of dissection.
D-dimer
While D-dimer seems like it might be appealing to help rule out the diagnosis in low risk patients, for such a rare diagnosis and poor test characteristics of D-dimer for dissection, guidelines do not recommend the use of D-dimer in the workup of aortic dissection.
For a deep dive into the world’s literature on D-dimer for aortic dissection visit EM Cases Journal Jam 9
POCUS for Aortic Dissection
Aortic dissection is the retinal detachment of the torso on POCUS.
While the sensitivity of POCUS by ED physicians to detect an intimal flap is only 67%, the specificity has been shown to be 99-100%. For patients suspected of the diagnsosis, look for an intimal flap that looks similar to a retinal detachment on POCUS and look for a pericardial effusion indicative of a retrograde dissection into the pericardium.
For an amazing case diagnosed on POCUS visit EDEblog
E) The Importance of the Correct Order & Aggressive Use of IV Medications in Aortic Dissection Management
First, call the CV surgeon for all aortic dissections (even for Type B dissections), as many Type B dissection require surgery. The indications for surgery in Type B dissection are: Malperfusion, ongoing progression, inability to control BP, and perforation.

1st: Pain control to counter sympathetic contribution to elevated heart rate and blood pressure
Fentanyl 25-50 mcg boluses
2nd: HR control to a goal of 60 bpm
Esmolol 0.5mg/kg bolus then 50-300 mcg/kg/min or
Labetolol 10-20mg bolus then 0.5-2mg/minor
Warning: giving a vasodilator without concomitant reduction in ionotropy may cause progression of dissection
3rd: BP control to a goal of SBP = 110
Nitroprusside 0.25-0.5 mcg/kg/min then titrate or
Nicardipine 5mg/hr
Warning: Beware of pseudohypotension! of the bilateral BPs use the higher BP reading.
For a detailed analysis of medication choices and arguments for esmolol as the first line medication visit EMCrit
Take Home Points for Aortic Dissection Diagnosis and Management
Remember the big pain pearls when taking a history: The 3 important questions, aortic dissection is the subarachnoid hemorrhage of the torso, migrating pain, colicky pain + opioids = badness and pain that comes and goes can still be a dissection.
Look for Marphan’s, listen for an aortic regurgitation murmur and feel for a pulse deficit.
Think not only about CP +1 but also 1+ CP
Know the CXR findings of loss or aortic knob/aortopulmonary window and the calcium sign, use POCUS to look for an intimal flap and pericardial effusion
Don’t be misled by a positive troponin
When it comes to management, treat pain first, then HR and then BP
For more on aortic dissection on EM Cases:
Episode 28: Aortic Dissection, Acute Limb Ischemia and Compartment Syndrome
Best Case Ever 13: Aortic Dissection
Journal Jam 9 – D-dimer to Rule Out Aortic Dissection
References
Hagan PG, Nienaber CA, Isselbacher EM, et al. The International Registry of Acute Aortic Dissection (IRAD): new insights into an old disease. JAMA. 2000;283(7):897-903. Full PDF
Rosman HS, Patel S, Borzak S, Paone G, Retter K. Quality of history taking in patients with aortic dissection. Chest. 1998;114(3):793-5.
Diercks D, Promes S, Schuur J, et al. Clinical Policy: Critical Issues in the Evaluation and Management of Adult Patients With Suspected Acute Nontraumatic Thoracic Aortic Dissection. Ann Emerg Med 2015;65:32-42.
Imamura H, Sekiguchi Y, Iwashita T, et al. Painless acute aortic dissection. Diagnostic, prognostic and clinical implications. Circ J. 2011;75(1):59-66.
Sobczyk D, Nycz K. Feasibility and accuracy of bedside transthoracic echocardiography in diagnosis of acute proximal aortic dissection. Cardiovasc Ultrasound. 2015;13:15.
Singer AJ, Hollander JE. Blood pressure. Assessment of interarm differences. Arch Intern Med. 1996;156(17):2005-8.
Fojtik J, Constantino T, Dean A. The Diagnosis of Aortic Dissection by Emergency Medicine Ultrasound. J Emer Med 2007; 32: 191-96.
Other FOAMed Resources on Aortic Dissection
EM Cases Journal Jam deep dive on D-dimer for aortic dissection
Basic review of aortic dissection EM Cases Episode 28 with David Carr and Anil Chopra
First10EM blog deep dive on D-dimer
EMCrit on management
CoreEM on the basics
emDocs on dissection
Now test your knowledge with a quiz.






Leave A Comment