While community acquired pneumonia (CAP) is ‘bread and butter’ emergency medicine, and the diagnosis is often a ‘slam dunk’, it turns out that up one third of the time, we are wrong about the diagnosis; that x-rays are not perfect; that blood work is seldom helpful; that not all antibiotics are created equal and that deciding who can go home and who needs to go to the ICU isn’t always so clear cut. With this in mind we are taking a deep dive into CAP with Dr. Leeor Sommer and Dr. Andrew Morris, from diagnosis to disposition so that we can better achieve our EM goals of stabilizing sick patients, getting the right diagnosis, initiating the best treatment with the information at hand, prognosticating/appropriately deciding on disposition of patients, and being healthcare and antimicrobial stewards…
Podcast: Play in new window | Download (Duration: 1:30:46 — 83.2MB)
Subscribe: Apple Podcasts
Podcast production, sound design & editing by Anton Helman; Script writing assistance by Andrew Cameron & Anton Helman.
Written Summary and blog post by Alexander Hartt and Anton Helman September, 2019
Cite this podcast as: Helman, A., Sommer, L., Morris, A. Episode 130 – Community Acquired Pneumonia – Emergency Management. Emergency Medicine Cases. September, 2019. https://emergencymedicinecases.com/community-acquired-pneumonia. Accessed [date]
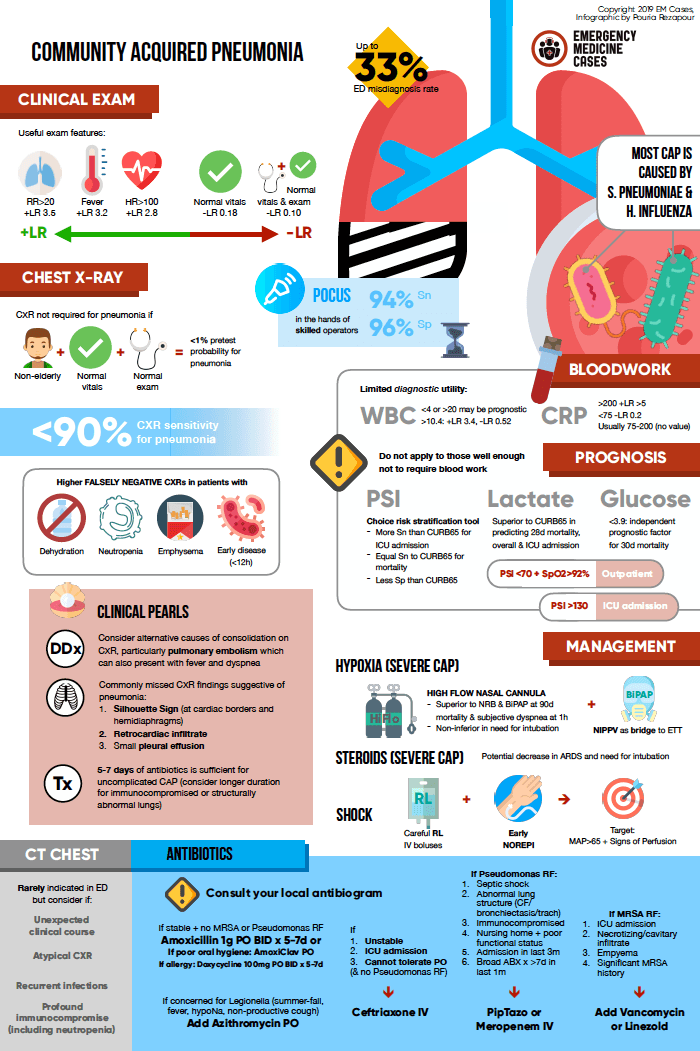
Sources of the high misdiagnosis rate of CAP in the ED
Some of the reasons why we misdiagnose CAP up to 1/3 of the time in the ED include pressure to make early treatment and disposition decisions (because of time-to-antibiotic “rules” in some jurisdictions), the expectation to have a definitive diagnosis when consulting services for admission, because the classic constellation of symptoms (cough, shortness of breath and fever) is often absent, there are many pneumonia mimics (CHF and PE being the most critical to identify and treat in the ED), early anchoring bias, there is overlap in clinical presentation of viral URI and pneumonia, blood tests may be misleading, CXR has poor accuracy, and there is no single historical or physical exam finding that has high enough likelihood ratio to shift pretest probability significantly.
To aid diagnostic accuracy and avoid over prescribing antibiotics, force yourself to consider the diagnosic criteria for CAP: fever, respiratory symptoms and imaging evidence of an infiltrate. Pay close attention to respiratory rate and oxygen saturation – the vast majority of patients with CAP will have an elevated respiratory rate and abnormal O2 sat, but don’t be fooled by the marathon runner – they can maintain perfectly normal vitals with their CAP.
Likelihood ratios for physical findings in CAP
The highest positive likelihood ratios of clinical findings for CAP from a 2019 meta-analysis are RR≥20 (3.47), fever (3.21) and HR>100 (2.79).
Normal vital signs combined with a normal pulmonary examination had a summary estimate -LR = 0.10 in a 2018 metaanalysis.
Normal vital signs alone have a -LR = 0.18 for CAP.
Pitfall: Using diagnosis of “acute bronchitis” in patients with viral respiratory illness, as it is a non-specific term that sets expectations by patients to be treated with antibiotics for a viral illness.
Blood tests for diagnosis and prognosis of CAP are promising statistically but usually not pragmatically useful
WBC > 10,400 per mm3 has +LR = 3.4, -LR = 0.52 for CAP, but normal values do not rule out pneumonia and WBC is not included in any of the prognostic decision tools. WBC in the extremes (<4, >20) may be of prognostic significance.
CRP of >200mg/L hav been found to have a +LR>5, while <75mg/L have been found to have a -LR<0.2, however, most patients will have values between these extremes, in which case there is little diagnostic or prognostic utility.
Procalcitonin may be a significantly better predictor for blood culture positivity in CAP than WBC count, C-reactive protein, and other clinical parameters, may reduce antibiotic exposure, and has been used to help guide cessation of antibiotic treatment, however procalcitonin does not appear to pragmatically change antibiotic exposure, LOS or mortality in the ED setting. Our experts recommend not ordering procalcitonin in the ED.
Hypoglycemia (blood glucose < 70 mg per dL or 3.89 mmol per L) at presentation is associated with increased 30-day mortality even after adjustment for other variables, including comorbid illness and Pneumonia Severity Index (PSI) score.
Lactate has been shown to be a better predictor of 28-day mortality, hospitalization and ICU admission than CURB-65 in ED CAP patients.
Chest x-ray indications, accuracy, false negatives and false positives for community acquired pneumonia
Indications for chest x-ray in patients with acute respiratory illness for CAP:
- At least one abnormal vital sign (Fever, tachycardia, RR>20)
- Two of: decreased breath sounds, crackles, absence of asthma
Common conditions that may lead to false negative chest x-ray in CAP
- Volume depletion
- Neutropenia
- Emphysema
- Early disease (first 12 hours)
Subtle chest x-ray findings that are often missed in CAP
- Silhouetting of heart border
- Small pleural effusion
- Retrocardiac infiltrate
Pitfall: Assuming that a non-apical lung infiltrate cannot be acute pulmonary tuberculosis
Indications for CT chest in suspected community acquired pneumonia
- Clinical course is not as expected (long duration, worsening despite appropriate CAP treatment)
- Recurrent infections
- An x-ray with atypical findings
- Neutropenic patients
- Profound immunocompromised state
While CT chest may be more accurate than chest x-ray at visualizing the upper lobes/lingula, showing interstitial edema of atypical pathogens, further characterizing necrotizing infection, multilobar disease, empyema, and pleural involvement, it is rarely indicated in the ED for suspected CAP.
POCUS perfomed by experienced operators is more accurate than chest x-ray for community acquired pneumonia, but is limited by the time it takes to perform
A 2014 meta-analysis concluded that, in the hands of experienced operators, ultrasound examination has a sensitivity and specificity as high as 94% and 96%, respectively. Ultrasound examination may offer an ideal alternative diagnostic modality in pediatric patients and critically ill patients in whom it is difficult to obtain a 2-view chest x-ray. However, for patients who are stable enough to go to the radiology department to get a 2 view chest x-ray, the time required to complete a thorough lung POCUS exam may be a limiting factor.
Indications for blood cultures in suspected community acquired pneumonia
- Severe CAP requiring ICU admission
- Evidence of sepsis
- Cirrhosis, asplenia or neutropenia
- Cavitary lesions or empyema on chest x-ray
Indications for sputum gram stain and cultures in suspected community acquired pneumonia
Patients with CAP are able to produce a valid sputum sample only 70% of the time. The sensitivity of sputum Gram stain ranges from 15% to 69% and specificity ranges from 11% to 100%. Many elderly patients with CAP are not able to produce an adequate specimen.
Consider sputum gram stain and cultures in the ED for patients with:
- Severe CAP
- Intubated patients with CAP
- History of alcohol abuse, liver disease, lung disease, leukopenia, cavitary infiltrates, asplenia, pleural effusion, and recent travel
Urine Legionella and Pneumococcus are rarely indicated in the ED
A 2009 retrospective analysis of Legionella CAP found 6 factors to be independent predictors:
- Fever (OR 1.67, p < 0.0001)
- Absence of sputum production (OR 3.67, p < 0.0001)
- Low serum Na (OR 0.89, p = 0.011)
- Elevated lactate (OR 1.003, p = 0.007)
- Elevated CRP (OR 1.006, p < 0.0001)
- Thrombocytopenia (OR 0.991, p < 0.0001)
Legionella may occur any time of year, but more illness is found in the summer and early fall.
Urine antigen testing in a 2009 meta-analysis showed a pooled sensitivity of 74% and specificity of 99% based on poor quality evidence.
In low prevalence areas, urine Legionella testing is not recommended in the ED by our experts as it is not cost effective.
Testing for Pneumococcal urine antigen in the ED is not recommended by our experts because empiric therapy will cover strep pneumococcus.
Antibiotic recommendations for community acquired pneumonia in Ontario
Consult your local biogram for recommendations in your area
- No risk factors for MRSA or pseudomonas, hemodynamically stable, non-ICU: amoxicillin or doxycycline (if penicillin allergy) or amoxicillin-clavulanic acid (if poor oral hygiene or non-ICU inpatient admission anticipated)
- No risk factors for MRSA or pseudomonas and are hemodynamically unstable, or have ICU admission planned, or are unable to tolerate oral antibiotics: IV ceftriaxone
- MRSA risk factors: add vancomycin or linezolid
- Pseudomonas risk factors: piperacillin-tazobactam or miropenem
The vast majority of pneumonias are caused by only 2 bacteria: Streptococcus pneumonia and Haemophilus influenzae. All strep pneumonia and most H flu are susceptible to penecillin or amoxicillin. Therefore,the first line antibiotic for CAP patients without risk factors for MRSA or pseudomonas and who are hemodynamically stable, based on a Cochrane review is amoxicillin 1g po bid.
For patients with true penicillin allergy doxycycline 100mg po bid is the recommended first line antibiotic for these with CAP. It has good atypical coverage and a low risk for C. diff.
Amoxacillin-clavulinic acid (Clavulin) does not appear to confer added coverage against strep pneumo (as the mechanism for resistance is not via beta-lactamase), however it can be considered as an alternative to amoxicillin or doxycycline in patients with poor oral hygiene and for non-ICU inpatients.
There is no consensus in the literature around adding azithromycin for atypical coverage. A 2014 JAMA article suggests that time to clinical stability favours the addition of azithromycin but there is no benefit for patient oriented outcomes. Likewise, the often mentioned anti-inflammatory properties of azithromycin does not confer benefit for patient oriented outcomes. In high prevalence Legionella regions, in patients with predictors (listed above) it is not unreasonable prescribe azithromycin in the ED.
Fluoroquinolones should not be first or even second line therapy. Serious adverse reactions include:
- Tendinopathy
- Multiple drug interactions
- Partial treatment of tuberculosis leading to diagnostic delay
- Increased risk of aortic dissection
Update 2021: Observational urgent care center study (across USA) of antibiotic prescribing rates for acute respiratory illnesses (ARIs) for patients <65 years old. Found patients of providers with high antibiotic prescribing rates received more future antibiotics after their initial index visit compared to patients of lower-prescribing providers. Receipt of antibiotics for ARI also increased likelihood of ARI visits in the future and ARI antibiotics received by their spouses. Abstract
Update 2022: A meta-analysis of six randomized controlled trials involving 834 patients comparing doxycycline to macrolides and fluoroquinolones for the treatment of non-severe community acquired pneumonia in adults found similar efficacy and adverse event rates, with lower length of hospitalization and lower cost of antimicrobial agent in the doxycycline group compared to levofloxacin group. Abstract
Oral antibiotics are as effective as IV antibiotics for most community acquired pneumonia
Oral antibiotics are recommended over IV antibiotics in the vast majority of ED patients with CAP or suspected CAP. There are at least 9 RCTs that show no clinical benefit for IV antibiotics over oral antibiotics for CAP. The bioavailablility of almost all antibiotics commonly used for CAP are comparable whether IV or po. In addition, IV antibiotics have a higher rate of side effects, and take longer time to administer in the ED.
IV antibiotics such as Cefriaxone are indicated in CAP patients who:
- Are hemodynamically unstable
- Have ICU admission planned
- Are unable to tolerate PO
MRSA is on the decline but should still be considered for at risk patients
Empiric therapy with vancomycin or linezolid for MRSA was recommended in 2011 IDSA guidelines for MRSA for hospitalized patients with severe community-acquired pneumonia defined by one of the following:
- A requirement for admission to the ICU
- Necrotizing or cavitary infiltrates
- Empyema
However, the prevalence of MRSA since 2011 has been declining with two 2016 studies finding a prevalence of only 0.7-3%.
MRSA nasal screening is of little value considering the low prevalence of MRSA CAP and the poor positive predictive value of the test. A meta-analysis of 22 studies with 5243 patients found that nasal screening had a pooled sensitivity of 70.9%, specificity of 90.3%, positive predictive value (PPV) of 44.8%, and negative predictive value (NPV) of 96.5% for MRSA pneumonia.
Indications for extended antibiotic coverage for Pseudomonas in community acquired pneumonia
Consider broadening antibiotic coverage to cover Pseudomonas with piperacillin-tazobactam, meropenem or ciprofloxacin in the setting of:
- Structurally abnormal lungs
- CF
- Bronchiectasis
- Tracheostomy
- Septic shock
- Broad spectrum antibiotics for >7 days in the last month
- Hospitalization for >1 day in last 3 months
- Immunocompromised (chemo, chronic steroids)
- Nursing home resident with poor functional status
It is unnecessary to provide antimicrobial coverage for anaerobes in patients suspected of aspiration pneumonia in the ED
Even in the setting of true aspiration pneumonia, the organisms involved are usually susceptible to beta-lactams (ceftriaxone). There is no need to add metronidazole or clindamycin.
Five-7 days duration of antibiotic treatment for uncomplicated community acquired pneumonia is sufficient
For uncomplicated CAP, 5-7 days of antibiotics is sufficient. More complex cases involving immunocompromised patients and those with structural lung disease will likely benefit from longer therapy. A metaanalysis of studies comparing treatment durations of 7 days or less with durations of 8 days or more showed no differences in outcomes and prospective studies have shown that 5 days of therapy are as effective as 10 days and 3 days are as effective as 8.
Treatment failure is defined by lack of defervenscence within 4-5 days and lack of subjective patient improvement within 72hrs
Our experts define treatment failure by a lack of defervescence within 4-5 days and lack of subjective patient improvement within 72hrs. Radiographic improvement can take weeks so is unreliable.
Adjunctive steroids may be of benefit in patients with severe community acquired pneumonia
Steroids are thought to curb the inflammatory response in CAP, reduce the frequency of acute respiratory distress syndrome, and decrease the length of illness. A systematic review and metaanalysis suggested that steroids reduce the need for mechanical ventilation and rate of acute respiratory distress syndrome by 5% with an NNT=20, however there are other high quality studies not reviewed in the metaanalysis that show no benefit. The SCCM/ESICM guidelines and expert consensus seems to favour steroids being reserved for those with severe CAP and those taking steroids chronically.
Initial steroid dosing options in the ED include:
- Dexamethasone 10mg IV
- Methylprednisolone 40mg IV
- Hydrocortisone 50mg IV
Learn more about steroid treatment in Journal Jam 17- Steroids for CAP and COVID Pneumonia
Update 2022: A meta-analysis of 16 RCTs including 3,842 hospitalized patients with community acquired pneumonia found no difference in all cause, in hospital mortality with corticosteroid therapy (RR 0.85, 95% CI 0.67 – 1.07), but was associated with a reduction in progression to mechanical ventilation (RR 0.51, 95% CI 0.33 – 0.77). Steroid use was also associated with subsequent readmission (RR 1.20, 95% CI 1.05 – 1.38). Abstract
Update 2024: A meta-analysis including 15 randomized controlled trials and 3,367 patients hospitalized with community acquired pneumonia found that all cause mortality was significantly lower in patients who received adjunctive corticosteroid therapy (6.2% vs 9.1%, RR 0.67, 95% CI 0.53-0.85). Corticosteroid therapy was also associated with reduced risk of ARDS (RR 0.24, P=0.002), reduction in time to clinical stability (1.5 days, P<0.001), with no associated increase in adverse events (RR 0.9, P=0.5). Abstract
Resuscitation of the patient with community acquired pneumonia and septic shock
Fluid choice and volume: Based on the SALT-ED and SMART trials, most experts agree that Ringer’s Lactate is the fluid of choice in septic shock patients. While massive fluid resucitation may worsen hypoxemia respiratory failure and precipitate need for intubation, under-resuscitation may worsen end-organ damage. Aiming for a MAP≥65 is a reasonable goal in addition to physical signs of end organ perfusion such as urine output and normal sensorium.
Norepinephrine indications and timing: Based on the CENSER trial it is reasonable to start peripheral norepinephrine as soon as the MAP <65 and/or there are signs of poor organ perfusion; the trial suggests that early norepinephrine in addition to fluid resuscitation results in less cardiogenic pulmonary edema, and possibly lower mortality.
The role of NIPPV in severe CAP: While NNPV theoretically theoretically prevents CAP patients from clearing secretions and mucus plugging, it may be used for limited periods of time as a bridge to intubation, and may be especially helpful in those with concurrent COPD exacerbation, where the evidence is clear for clinical benefit.
Learn more about COPD and pneumonia in Episode 24: COPD & Pneumonia
The role of High-Flow Nasal Cannula (HFNC) in severe CAP: The FLORALI trial suggested that HFNC may improve 90 day survival as well as subjective dyspnea and respiratory discomfort at 1-hr compared to non-rebreather and BiPAP in severely hypoxic CAP patients. It also showed that HFNC is non-inferior to non-rebreather facemask and BiPAP for reducing the need for intubation. HFNC is thought to reduce the work of breathing prior to respiratory exhaustion.
Pneumonia Severity Index (PSI) is the risk stratification tool of choice for community acquired pneumonia
PSI is more sensitive than SMART-COP and much more sensitive than CURB-65 for determining which patients will require ICU admission, while offering equal sensitivity for mortality for CAP overall. Despite CURB-65 having a higher specificity for CAP than PSI, our experts recommend PSI as the risk stratification tool of choice.
Problems with PSI
- PSI may underestimate the severity of pneumonia in an otherwise young healthy patients
- PSI does not include psychosocial conditions or cognitive impairments that may preclude discharge from the ED
- Any patient over 50 years of age is automatically classified as risk class 2 which may exaggerate their risk
Note that all CAP risk stratification tools rely on blood work so they do not apply to those well enough to not get lab testing.
Validated IDSA/ATS criteria for ICU admission
Patients with 3 or more criteria may benefit from ICU admission:
- Respiratory rate >29 breaths/min
- Hypotension requiring volume resuscitation
- PaO2/FiO2 < 250 (patients requiring >3 liters oxygen)
- Temperature < 36C
- Confusion
- Multilobar infiltrates
- BUN >20 mg/dL
- WBC <4,000/mm3
- Platelets <100,000/mm3
In addition, multilobar pneumonia is an independent risk factor for increased mortality in CAP.
Discharge criteria for outpatient care in patients with community acquired pneumonia
| PSI Score | Risk | Disposition |
|---|---|---|
| ≤70 | Low risk | Outpatient care |
| 71-90 | Low risk | Outpatient vs. Observation admission |
Some experts recommend a CURB-65 score of zero as criteria for outpatient care, however CURB-65 was validated as mortality prediction tool, and was not designed to determine disposition. Our experts recommend the following minimal criteria for outpatient care of CAP:
- RR<24
- O2sat ≥92%
- Normal mental status
- Able to tolerate oral intake
- Psychosocial support
Oxygen saturations less than 92% are associated with major adverse events in outpatients with CAP.
Take home points for emergency management of community acquired pneumonia
- We often misdiagnose CAP. To help us be better diagnosticians think about the diagnostic criteria (fever, respiratory symptoms and imaging evidence of infiltrate) and pay close attention to respiratory rate and oxygen saturation– the vast majority of patients with CAP will have an elevated respiratory rate and abnormal O2 sat, but don’t be fooled by the marathon runner – they can maintain perfectly normal vitals with their CAP.
- Not all cough is CAP. Think about the differential so that you do not overdiagnose CAP and you do not miss other important diagnoses.
- Not all patients in the ED with cough require a chest x-ray. Indications for chest x-ray include: At least one abnormal vital sign (fever, tachycardia, RR>20) and 2 of decreased breath sounds, crackles or absence of asthma. If you do get a chest x-ray remember that they can be normal or near normal early on the clinical course and in severely dehydrated or immunocompromised patients. Consider the differential diagnosis when you see an infiltrate – don’t just assume CAP.
- Procalcitonin has little, if any, role in the ED but may be useful for the inpatient team in predicting prognosis and duration of therapy. While WBC is usually unrevealing, extremes of WBC can help risk stratify your patients.
- For the septic CAP consider high flow nasal oxygen and/or NIPPV as a bridge to intubation; fluid resuscitation requires a delicate balance of considerations – don’t just slam in a few litres of crystalloid, but at the same time be sure not to under-resuscitate. Start peripheral norepinephrine early in CAP patients with septic shock – as soon as the MAP<65.
- For antibiotic choices, consult your local biogram. In Ontario, the recommended first-line outpatient therapy for CAP is currently amoxicillin 1g po bid. The recommended first-line inpatient non-ICU therapy for CAP is now amoxicillin-clavulanate 875mg/125mg po bid or ceftriaxone 1g iv q24h. Consideration for adding azithromycin empirically should only be given during the months of June through October to cover Legionella, MRSA only for those at risk (the prevalence is declining) and coverage for pseudomomas only in at risk patients. Oral antibiotics and as effective as IV antibiotics for the majority of patients with CAP.
- Recommended duration of therapy for most CAP is 5-7 days. Know the exceptions.
- PSI is the risk stratification tool of choice. Low-risk patients suitable for discharge from the ED should be defined by a PSI ≤ 70 and an oxygen saturation of at least 92% on room air. Use a PSI >130 as criteria for ICU admission.
References
- Ruiz-gonzález A, Utrillo L, Bielsa S, Falguera M, Porcel JM. The Diagnostic Value of Serum C-Reactive Protein for Identifying Pneumonia in Hospitalized Patients with Acute Respiratory Symptoms. J Biomark. 2016;2016:2198745.
- Htun TP, Sun Y, Chua HL, Pang J. Clinical features for diagnosis of pneumonia among adults in primary care setting: A systematic and meta-review. Sci Rep. 2019;9(1):7600.
- Marchello, C et al. Signs and symptoms that rule out community-acquired pneumonia in outpatient adults: a systematic review and meta-analysis. JABFM. 2018; 32;2;234-247.
- Mortensen EM, Garcia S, Leykum L, Nakashima B, Restrepo MI, Anzueto A. Association of hypoglycemia with mortality for subjects hospitalized with pneumonia. Am J Med Sci. 2010;339(3):239–243.
- Müller F, Christ-Crain M, Bregenzer T, et al.; ProHOSP Study Group. Procalcitonin levels predict bacteremia in patients with community-acquired pneumonia: a prospective cohort trial. Chest. 2010;138(1):121–129.
- Christ-Crain M, Stolz D, Bingisser R, et al. Procalcitonin guidance of antibiotic therapy in community-acquired pneumonia: a randomized trial. Am J Respir Crit Care Med. 2006;174(1):84–93.
- Chen YX, Chun-Shen L. Lactate on emergency department arrival as a predictor of mortality and site-of-care in pneumonia patients: a cohort study. Thorax 2015;1–7.
- Ebell MH. Predicting pneumonia in adults with respiratory illness. Am Fam Physician. 2007;76(4):560-2.
- McNally, M., Curtain, J., O’Brien, K. K., Dimitrov, B. D. & Fahey, T. Validity of British Thoracic Society guidance (the CRB-65 rule) for predicting the severity of pneumonia in general practice: systematic review and meta-analysis. The British journal of general practice : the journal of the Royal College of General Practitioners 60, e423-433, doi:10.3399/bjgp10X532422 (2010).
- Majumdar, S. R., Eurich, D. T., Gamble, J. M., Senthilselvan, A. & Marrie, T. J. Oxygen saturations less than 92% are associated with major adverse events in outpatients with pneumonia: a population-based cohort study. Clin Infect Dis 52, 325-331, doi:10.1093/cid/ciq076 (2011).
- Holter, J. C. et al. Etiology of community-acquired pneumonia and diagnostic yields of microbiological methods: a 3-year prospective study in Norway. BMC Infect Dis 15, 64, doi:10.1186/s12879-015-0803-5 (2015).
- Jain, S. et al. Community-Acquired Pneumonia Requiring Hospitalization among U.S. Adults. N Engl J Med 373, 415-427, doi:10.1056/NEJMoa1500245 (2015).
- Lidman C, Burman LG, Lagergren A, ÖrtQvist Å. Limited value of routine microbiological diagnostics in patients hospitalized for community-acquired pneumonia. Scand J Infect Dis 2002;34:873-879.
- Pakhale, S. et al. Antibiotics for community-acquired pneumonia in adult outpatients. Cochrane Database Syst Rev 10, CD002109, doi:10.1002/14651858.CD002109.pub4 (2014).
- Staub LJ, Mazzali biscaro RR, Kaszubowski E, Maurici R. Lung Ultrasound for the Emergency Diagnosis of Pneumonia, Acute Heart Failure, and Exacerbations of Chronic Obstructive Pulmonary Disease/Asthma in Adults: A Systematic Review and Meta-analysis. J Emerg Med. 2019;56(1):53-69.
- Gadsby, N. J. et al. Comprehensive Molecular Testing for Respiratory Pathogens in Community-Acquired Pneumonia. Clin Infect Dis 62, 817-823, doi:10.1093/cid/civ1214 (2016).
- Postma, D. F. et al. Antibiotic treatment strategies for community-acquired pneumonia in adults. N Engl J Med 372, 1312-1323, doi:10.1056/NEJMoa1406330 (2015).
- Garin, N. et al. beta-Lactam monotherapy vs beta-lactam-macrolide combination treatment in moderately severe community-acquired pneumonia: a randomized noninferiority trial. JAMA Intern Med 174, 1894-1901, doi:10.1001/jamainternmed.2014.4887 (2014).
-
Aliberti S, Reyes LF, Faverio P, et al. Global initiative for meticillin-resistant Staphylococcus aureus pneumonia (GLIMP): an international, observational cohort study. Lancet Infect Dis. 2016;16(12):1364-1376.
- Self WH, Wunderink RG, Williams DJ, et al. Staphylococcus aureus Community-acquired Pneumonia: Prevalence, Clinical Characteristics, and Outcomes. Clin Infect Dis. 2016;63(3):300-9.
- Liu C, Bayer A, Cosgrove SE, et al. Clinical practice guidelines by the Infectious Diseases Society of America for the treatment of methicillin-resistant Staphylococcus aureus infections in adults and children. Clin Infect Dis. 2011;52(3):e18–55.
- Siemieniuk, R. A. et al. Corticosteroid Therapy for Patients Hospitalized With Community-Acquired Pneumonia: A Systematic Review and Meta-analysis. Ann Intern Med 163, 519-528, doi:10.7326/M15-0715 (2015).
- Li, J. Z., Winston, L. G., Moore, D. H. & Bent, S. Efficacy of short-course antibiotic regimens for community-acquired pneumonia: a meta-analysis. Am J Med 120, 783-790, doi:10.1016/j.amjmed.2007.04.023 (2007).
- Dimopoulos, G. et al. Short- versus long-course antibacterial therapy for community-acquired pneumonia : a meta-analysis. Drugs 68, 1841-1854 (2008).
- Choudhury, G. et al. Seven-day antibiotic courses have similar efficacy to prolonged courses in severe community-acquired pneumonia–a propensity- adjusted analysis. Clin Microbiol Infect 17, 1852-1858, doi:10.1111/j.1469- 0691.2011.03542.x (2011).
- Avdic, E. et al. Impact of an antimicrobial stewardship intervention on shortening the duration of therapy for community-acquired pneumonia. Clin Infect Dis 54, 1581-1587, doi:10.1093/cid/cis242 (2012).
- Uranga, A. et al. Duration of Antibiotic Treatment in Community-Acquired Pneumonia. JAMA Internal Medicine, doi:10.1001/jamainternmed.2016.3633 (2016).
- Burnham JP, Kakol MK, Vazquez guillamet MC. Methicillin-resistant Staphylococcus aureus Nasal Screening Adds Limited Value to the Choice of Empiric Antibiotics in Community-acquired Pneumonia. Clin Infect Dis. 2019;68(7):1251.
- Welker JA et al. Antibiotic timing and errors in diagnosing pneumonia. Arch Intern Med 2008 Feb 25; 168:351.
- Musher DM, Thorner AR. Community-acquired pneumonia. N Engl J Med 2014;371:1619–28.
- Waterer GW, Rello J, Wunderink RG. Management of community-acquired pneumonia in adults. Am J Respir Crit Care Med 2011;183:157–64.
- Siemieniuk RAC, Meade MO, Alonso-Coello P, et al. Corticosteroid therapy for patients hospitalized with community-acquired pneumonia. A systematic review and meta-analysis. Ann Intern Med 2015;163:519–28.
- Chalmers JD. Corticosteroids for community-acquired pneumonia: a critical view of the evidence. Eur Respir J 2016;48:984–6.
- Chavez MA, Shams N, Ellington LE, et al. Lung ultrasound for the diagnosis of pneumonia in adults: a systematic review and meta-analysis. Respir Res 2014;15:50. 33.
- Chen YX, Chun-Shen L. Lactate on emergency department arrival as a predictor of mortality and site-of-care in pneumonia patients: a cohort study. Thorax 2015;1–7.
- Mandell LA, Wunderink RG, Anzueto A, et al. Infectious Diseases Society of America/American Thoracic Society consensus guidelines on the management of community-acquired pneumonia in adults. Clin Infect Dis 2007;44(S2):S27–72.
- Fiumefreddo R, Zaborsky R, Haeuptle J, et al. Clinical predictors for Legionella in patients presenting with community-acquired pneumonia to the emergency department. BMC Pulm Med. 2009;9:4.
- Aujesky D, Fine MJ. The Pneumonia Severity Index: a decade after the initial derivation and validation. Clin Infect Dis 2008;47:S133–9.
- Charles PG, Wolfe R, Whitby M, et al. SMART-COP: a tool for predicting the need for intensive respiratory or vasopressor support in community-acquired pneumonia. Clin Infect Dis 2008;47(3):375–84.
- Lee M, Oh J, Kang C, et al. Guideline for Antibiotic Use in Adults with Community-acquired Pneumonia. Infect Chemother. 2018;50(2):160-198.
- Cretikos M, Bellomo R, Hillman K, Chen J, Finfer S, Flabouris A. Respiratory rate: the neglected vital sign. Med J Aust. 2008;188(11):657-659.
- Chalmers J, Taylor J, Mandal P, et al. Validation of the Infectious Diseases Society of America/American Thoratic Society minor criteria for intensive care unit admission in community-acquired pneumonia patients without major criteria or contraindications to intensive care unit care. Clin Infect Dis. 2011;53(6):503-511.
- Williams J, Greenslade J, Chu K, Brown A, Lipman J. Utility of community-acquired pneumonia severity scores in guiding disposition from the emergency department: Intensive care or short-stay unit? Emerg Med Australas. 2018;30(4):538-546.
- Frat JP, et al. High-flow oxygen through nasal cannula in acute hypoxemic respiratory failure. NEJM. 2015. 372(23):2185-2196.
- Pastores S, Annane D, Rochwerg B, Corticosteroid G. Guidelines for the diagnosis and management of critical illness-related corticosteroid insufficiency (CIRCI) in critically ill patients (Part II): Society of Critical Care Medicine (SCCM) and European Society of Intensive Care Medicine (ESICM) 2017. Intensive Care Med. 2018;44(4):474-477.
- Musher DM, Roig IL, Cazares G, Stager CE, Logan N, Safar H. Can an etiologic agent be identified in adults who are hospitalized for community-acquired pneumonia: results of a one-year study. J Infect 2013;67:11-8
- Abers MS, Musher DM. Clinical prediction rules in community-acquired pneumonia: lies, damn lies and statistics. QJM 2014;107:595-6.
- Li JZ, Winston LG, Moore DH, Bent S. Efficacy of short-course antibiotic regimens for community-acquired pneumonia: a meta-analysis. Am J Med 2007;120: 783-90.
- Dunbar LM, Khashab MM, Kahn JB, Zadeikis N, Xiang JX, Tennenberg AM. Efficacy of 750-mg, 5-day levofloxacin in the treatment of community-acquired pneumonia caused by atypical pathogens. Curr Med Res Opin 2004;20:555-63. [Erratum, Curr Med Res Opin 2004;20:967.]
- el Moussaoui R, de Borgie CA, van den Broek P, et al. Effectiveness of discontinuing antibiotic treatment after three days versus eight days in mild to moderate-severe community acquired pneumonia: randomised, double blind study. BMJ 2006;332:1355.
- Fine MJ. et. al. A prediction rule to identify low-risk patients with community-acquired pneumonia. N Engl J Med. 1997 Jan 23;336(4):243-50.
- Shah BA, et. al. Validity of Pneumonia Severity Index and CURB-65 Severity Scoring Systems in Community Acquired Pneumonia in an Indian Setting. The Indian Journal of Chest Diseases & Allied Sciences. 2010;Vol.52.
Other FOAMEd resources on Community Acquired Pnuemonia
https://emcrit.org/ibcc/pneumonia/
Community Acquired Pneumonia – (LITFL CCC, Chris Nickson)
Evidence-based treatment for severe community-acquired pneumonia (PulmCrit)
Radiologic – Ultrasonic – Pathologic correlation for pneumonia (PulmCrit)
Antibiotics
Which patients with pneumonia need MRSA coverage? (PulmCrit)
Update in community acquired pneumonia: Macrolide resistance (Anand Swaminathan, Rebel EM)
Six reasons to avoid fluoroquinolones in the critically ill (PulmCrit)
Modes of noninvasive support
Pneumonia, BiPAP, secretions, and HFNC: Lessions learned from FLORALI trial (PulmCrit)
Mastering the dark arts of BiPAP & HFNC (PulmCrit)
Metabolic therapies
Steroid for community-acquired pneumonia (PulmCCM, Jon-Emile Kenny)
Corticosteroids for pneumonia: Ready for primetime? (emDocs, Brit Long)
Metabolic resuscitation for severe pneumonia? (PulmCrit)
POCUS for pneumonia
Pneumonia: Five minute sono (Jacob Avila)
Pneumonia US library (LITFL, James Rippey)
Drs. Helman, Sommer and Morris have no conflicts of interest to declare





Hi,
my name is Felix. I am working as an ED physician in Germany and enjoy listening to your podcast. Please keep up that fantastic work!
Relating to your current episode of CAP I would like to mention two points.
1- I am very glad that you provide good evidence for treating patients orally. However in our healthcare system we are often kind of forced to go for iv antibiotics for inpatients. The health insurance declines paying the hospital bill if the patient did not receive iv medication postulating the stay was not necessary.
2- You were quite sceptic about the use of CRP/leucocyte count/pct because high values do exist in viral infections as well. However I think if someone has markedly elevated levels it still means that there is some important inflammation going on. So in an ill appearing patient with high inflammatory markers I would then treat with antibiotics because of the possible (threatening) bacteremia and take the risk of unnecessary treatment and possible adverse events of the drug in case of a viral cause.
I hope you can follow my reasoning although I am lacking some fluency in English…
Thanks again for you great podcasts – I am looking forward to the next episode.
Felix
In uncertainty a CAT scan of the chest is more accurate .
Further PCR is developing rapidity of diagnosis eg TB and there are faster tests for drug sensitivity emerging .
Was not totally clear when, if ever, it is indicated to give fluoroquinolones (levofloxacin, moxifloxacin) PO for CAP in the outpatient setting. Is it only recommended if there are contraindications to amoxicillin or doxycycline? It appears where I work (California) that we are more apt to give Levofloxacin to elderly with CAP. Perhaps there is a belief that it’s better.than amoxicillin. Thanks.
For outpatient Amoxicillin is first line and if allergy to Penicillin then Doxycycline regardless of age. FDA has warned that fluoroquinolones are associated with Aortic Dissection, so probably best to avoid them in the elderly, especially if history of hypertension or AAA. While fluoroquinolones are in the newest guidelines as a choice for “severe” CAP, the evidence is weak that they are any better than first line agents for outpatient CAP. For severe CAP, ceftriaxone +/- azithro (for at risk patients) is a better choice.
The question :What distinguishes bronchitis from pneumonia was not answered .
Looking Sick
A lot of coughing with phlegm
Aches and Pains
Increased respiratory rate above 20
low po2 at 94 and below
higher fever 39.0C and in severer cases
tachcardia PR 110 or greater
hypotension BP pressure systolic 90 or less
respiratory rate 24 and over and
Po2 on air by pulse Oximeter 92 and below
Would be alerting .
Hi thanks for the excellent podcast. In the U.K. we use CURB score not only to help decide whether to admire but also use for treatment guidance.
You didn’t mention Mycoplasma pneumonia – when to suspect and what treatment to add.
HI
I am an Advanced Nurse Practitioner working in a Rual Access Hospital without on site supervision. I love these keeps me up to date and is fast and easy reads.
Thank you so much for keeping it free.
Karla