In this episode on Pulmonary Embolism we have the triumphant return of Dr. Anil Chopra, the Head of the Divisions of Emergency Medicine at University of Toronto, and Dr. John Foote the CCFP(EM) residency program director at the University of Toronto. We kick it off with Dr. Foote’s approach to undifferentiated dyspnea and explanation of Medically Unexplained Dyspea (‘MUD’) and go on to discuss how best to develop a clinical pretest probability for the diagnosis of pulmonary embolism using risk factors, the value of the PERC rule, Well’s criteria and how clinical gestalt plays into pre-test probability. Dr. Chopra tells about the appropriate use of D-dimer to improve our diagnostic accuracy without leading to over-investigation and unwarranted anticoagulation. We then discuss the value of V/Q scan in the workup of PE, and the pitfalls of CT angiography. A discussion of anticoagulation choices follows and the controversies around thrombolysis for submassive PE closes the podcast. Dr. Chopra & Dr. Foote answer such questions as: What are the most important under-recognized risk factors for pulmonary embolism, and how does the absence of risk factors change the pre-test probability of PE? Should we be using D-dimer in pregnant patients? How does the D-dimer value change with age and how can we incorporate this into our work-up? How does an experienced doctor’s clinical gestalt compare to clinical desicion rules in working up PE? In what situations is V/Q scan preferred over CT angiogram for the diagnosis of PE? How should we manage patients with subsegmental PE found on CTA? What is the danger of aggressive fluid resuscitation in patients with massive PE? Which patients can be sent home from the ED with PE? Should we thrombolyse patients with submassive PE? What is Medically Unexplained Dyspnea (MUD) and how can we identify these patients and avoid lengthy work-ups?
Written summary and blog post prepared by Claire Heslop, edited by Anton Helman March 2012
Cite this podcast as: Chopra, A, Foote, J, Helman, A. Pulmonary Embolism. Emergency Medicine Cases. March, 2012. https://emergencymedicinecases.com/episode-21-respiratory-emergencies-1-pulmonary-embolism/. Accessed [date].
APPROACH TO THE PATIENT WITH DYSPNEA
Big Categories to Consider
- Respiratory
- Cardiac
- The most common presentation of MI in patients >85 y.o. is dyspnea
- Effusion and tamponade may also present as dyspnea
- Anemia (Hb < 80)
- Neuromuscular (E.g. Guillan Barre)
- Metabolic acidosis of any cause
COPD patients are at higher risk than the general population for PE, CHF, pneumonia, tamponade, pneumothorax, and MI
- Rizkallah and colleagues found 20% of COPD exacerbation admissions had PE
- Suspect other processes if your patient with COPD presents atypically from their usual episodes, or if they are not improving with medical therapies for their exacerbation
Medically Unexplained Dyspnea (‘MUD’)
A condition characterized by a sensation of a need to take a deep inspiration or the feeling of an ‘oppressive’ chest plus symptoms of anxiety, in the absence of wheeze, cough, or sputum, ECG, CXR, or peak flow abnormalities, and without cardiopulmonary explanations for their dyspnea. It has been called “the IBS of respiratory”. Patients are often over-investigated without significant findings. A diagnosis of MUD should not be made on a single ED visit, but should be considered in patients who have had extensive previous work-up for the same symptoms in the past.
PULMONARY EMBOLISM WORK-UP & MANAGEMENT
Risk Factors and Symptoms for Pulmonary Embolism
Classic Risk Factors for PE: “THROMBOSIS”
Trauma/Travel
Hypercoagulable state/HRT
Recreational drugs (IVDU)
Older
Malignancy
Birth Control
Obesity/Obstetrical (The risk of Pulmonary Embolism is highest in the first 6 weeks postpartum)
Surgery
Immobilization
Sickness
Chronic illnesses such as Lupus, CAD, CHF & COPD contribute to risk.
Varicose veins are a risk factor for Pulmonary Embolism, but when they generate clots, they may be less likely to embolize
Note: 20% of Pulmonary Embolism patients will have no identifiable risk factors at presentation.
Suspect PE when you see RAPID onset of symptoms, dyspnea+/- tachypnea, and pleuritic chest pain. Always be alert to older patients who present atypically.
Update 2017: Prospective data from this Lancet cohort study led authors to conclude the YEARS algorithm approach to PE management resulted in an absolute 14% decrease in CTPA examinations amongst all ages. While promising, the study requires further external validation. Abstract
D-dimer in the work-up of Pulmonary Embolism
Overuse of the D-dimer can lead to over-investigation and anticoagulation, unless used wisely and in the correct clinical context.
Tips for using D-Dimer wisely:
- use a high sensitivity assay,
- test low risk patients,
- test when you expect it will be negative (to rule out PE),
- don’t test D-dimer if you plan to order a CT regardless
Is D-dimer useful in pregnancy?
- The American Thoracic Society Guidelines state D-dimer should not be used to rule out Pulmonary Embolism in pregnancy.
- D-dimer rises in the 2nd and 3rd trimester, and stays high post-partum.
- Cutoffs of 1.5x, 2x and 2.2x the local threshold per trimester have been suggested. **However, our experts suggest that only in the first trimester a negative D-dimer may be useful to rule out PE.**
- Guidelines for investigating for Pulmonary Embolism in pregnancy start with evaluation for signs & symptoms of DVT. If these are found, a leg doppler is done.
- If no DVT is suspected or found, order a CXR:
- CXR normal? ->V/Q scan* CXR abnormal? ->CT scan
- *V/Q scan exposes pregnant women to less radiation than a CT scan for PE (V/Q and CT expose the fetus to similar radiation dosages; both dosages are low and well below background levels.)
What about the elderly?
- Consider adjusting the upper limit cutoff for negative D-dimer in patients older than 50
- Adjusted upper limit = age x 10
What about a patient on Warfarin?
- D-dimer can be falsely negative due to anticoagulation
Update 2022: A prospective study examining a new modified age-adjusted clinical decision rule “Adjust-Unlikely” for pulmonary embolism including 1703 patients found a sensitivity of 100% and specificity of 32.4%. YEARS had a higher specificity of 45%, but missed 1 in 14 cases of PE. “Adjust-Unlikely” used the age-adjusted D-Dimer (age x 10 in patients older than 50) in cases were PE was not the most likely diagnosis, but used the standard D-Dimer level of 500ng/mL in those who PE was the most likely diagnosis. Abstract
Well’s criteria for pretest probability of pulmonary embolism
Use Well’s criteria (3) to assess pretest probability for Pulmonary Embolism:
Clinical signs of symptoms of DVT +3 points
PE is #1 Diagnosis or equally likely +3 points
Heart rate > 100 +1.5 points
Immobilization at least 3 days, or surgery in the previous 4 weeks +1.5 points
Previous, objectively diagnosed PE or DVT +1.5 points
Hemoptysis +1 point
Malignancy with treatment within 6 months, or palliative +1 point
If the patient is determined to be low risk (
Alternatively consider a rule-out criteria such as PERC (see below).
If the D-dimer is negative consider stopping the workup.
If the D-dimer is positive consider chest CTA.
If the patient is determined to be moderate risk (score 2-6 points, 16.2% incidence of PE), consider high sensitivity D-dimer testing or chest CTA.
If the D-dimer is negative consider stopping workup.If the D-dimer is positive consider chest CTA.
If the patient is determined to be high risk (score >6 points: 37.5% incidence of PE) consider chest CTA. D-dimer testing is not recommended.
Calculate Well’s Score on MD Calc
Update 2017: A recent study has evaluated the usage of interval likelihood ratios to determine different D-dimer cut offs for ordering a CTPA, depending on one’s pretest probability for pulmonary embolism. For instance, if one has a lower pretest probability for PE (5%) by Well’s Criteria, a higher D-dimer cut off (<1000ng/mL) could be accepted as “negative”. Alternatively, if an individual has a higher pretest probability (15%), they would have a lower D-dimer cutoff (<500ng/mL) to consider PE ruled out. Abstract
Update 2019: EM Quick Hits 23 on Clinical Probability Adjusted D-Dimer: The PEGeD Study
PERC Rule to help decide which low risk patients need a work-up for pulmonary embolism
The “Pulmonary Embolism Rule-Out Criteria” (PERC) is a step 2 decision support for excluding PE. If none of the eight PERC features are present in a patient that has been deemed low risk by Well’s criteria or by clinical gestalt, then no further tests are indicated.
PERC RULE: “HAD CLOTS”
Hormones (estrogen)
Age >50
DVT or PE history
Coughing up blood (hemoptysis)
Lower extremity swelling
O2 <95%
Tachycardia
Surgery or trauma in past 4 weeks
The PERC rule is only accurate when applied to LOW risk patients.
Calculate PERC score on MD Calc
Update 2021: Prospective study across 6 ED’s including 935 patients, assessing clinical deterioration or death within 5 days of pulmonary embolism (PE) diagnosis using PE-SCORE criteria. Primary outcome was death or deterioration (respiratory failure, cardiac arrest, dysrhythmia, sustained hypotension, and rescue reperfusion intervention). PE-SCORE resulted in a score of zero (low-risk) with a negative predictive value of 97.9% and 2% had the primary outcome, and a score >5 to define high-risk patients who all had the primary outcome. Abstract
Update 2022: A post hoc analysis of two European prospective cohorts including 2968 patients presenting to the ED with low clinical probability of pulmonary embolism found that the removal of hemoptysis from the PERC, YEARS and PEGeD clinical decision tools did not significantly alter the the performance of these clinical decision tools or lead to a clinically significant reduction in diagnostic imaging. Abstract
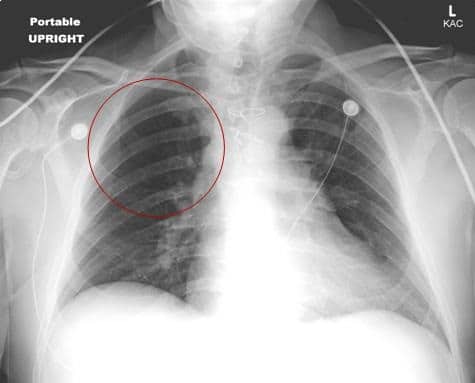
What are the Chest X-Ray Findings for Pulmonary Embolism?
Although the CXR may appear unremarkable, about 75% of patients with Pulmonary Embolism have findings resulting from the clot, such as an elevated hemi-diaphragm, a unilateral pleural effusion, enlarged pulmonary arteries, and even infiltrates.
In young patients with pleuritic chest pain, a pleural effusion increases the likelihood of PE.
Uncommon findings of Pulmonary Embolism (see below) have low sensitivity for detecting PE.
Uncommon findings for Pulmonary Embolism include:
- “Westermark’s Sign” representing a region of oligemia
- Wedge shaped consolidation aka “Hampton’s Hump”
ECG Findings in Pulmonary Embolism
ECG is useful to rule out other diseases, and~30% will be normal. ECG findings that may suggest PE include:
- sinus tachycardia
- RV strain pattern
- incomplete RBBB
- flipped T waves in anterior leads
- S1Q3T3 (poor sensitivity & specificity)
- flipped T waves in anterior and inferior leads, an uncommon finding which has been shown to be highly specific for PE
Update 2022: A prospective, multicenter study of 1629 newly confirmed PE patients found that supraventricular tachycardia was the only ECG finding with a significant OR for clinical deterioration defined as respiratory failure, hypotension, dysrhythmia, cardiac arrest, and escalated PE (OR 2.87). However, T-wave inversions in V2-4, incomplete RBBB, ST elevation in aVR, sinus tachycardia and S1-Q3-T3 were all independent predictors of abnormal RV function on echocardiogram. Abstract
Do all patients with Pulmonary Embolism patients require admission?
Aujesky and colleagues reported outpatient management for PE was not inferior to management in hospital, for hemodynamically stable patients with no significant comorbidity, who could be safely anticoagulated, and closely followed as outpatients.
Once diagnosed, PE therapy begins with anticoagulation. LMWH (Fragmin or Enoxaparin) or Fondaparinux for at least 5 days and the INR=2.0-3.0, plus Warfarin
Update 2016: NOACs for treatment of PE
NOACs: Dabigatran (Pradaxa®), Rivaroxaban (Xarelto®), Apixaban (Eliquis®)
These NOACs have been shown in large studies to be effeictive for the initial (rivaroxaban, apixaban), acute (all agents) and extended (all agents) treatment of PE.Dosages of NOACs for Pulmonary Embolism
Rivaroxaban 15 mg bid x 21 days, followed by 20 mg once daily for at least 3 months
Apixaban 10 mg bid x 7 days, followed by 5 mg bid
Note that 5 days of therapeutic LMWH should be administered prior to the initiation of Dabigatran when this anticoagulant is used for the treatment of PE.
Update 2018: A prospective, randomized controlled trial of 114 patients showed low-risk ED pulmonary embolism patients (as per Hestia criteria) who received rivaroxaban and were discharged from the ED, had similar outcomes to standard of care, shorter hospital stay and lower costs over 30 days. Abstract
Update 2022: Multi-center prospective cohort study across 18 sites from 2011-2021 including 292 patients with subsegmental pulmonary embolism (PE) (without proximal deep venous thrombosis), managed without anticoagulation. Found recurrent venous thromboembolism in 3.1% of patients during 90-day follow-up period, with no fatal recurrent PE. Excluded patients who were hospitalized, pregnant, had active cancer, history of PE/DVT or hypoxia. Abstract
Massive and Submassive Pulmonary Embolism
Massive Pulmonary Embolism: acute PE with sustained hypotension (systolic blood pressure 15 minutes or requiring inotropic support, not due to another cause), pulselessness, or profound bradycardia.
Thrombolysis is indicated in massive PE.
Submassive Pulmonary Embolism: no sustained hypotension, but either RV dysfunction (based on echocardiography, CT, BNP, Tropinin or ECG evidence) and/or myocardial necrosis (based on troponin elevation).
Our experts do not recommend thrombolysis in submassive PE. However, some experts believe that lytics are indicated if there is evidence of present or developing circulatory or respiratory insufficiency; or moderate to severe RV injury.
Finally: Fluid resuscitation in Pulmonary Embolism with shock is indicated as first line support, but *resuscitate judiciously* as volume expansion may impair RV function.
Update 2017: A recent RCT found that thrombolytic therapy for submassive pulmonary embolism did not provide a mortality benefit, nor a change in dyspnea scores or the development of pulmonary hypertension at 3 years post-treatment. Abstract
For Dr. Joel Yaphe’s interpretation of the literature on submassive pulmonary see Episode Episode 61 Whislter’s Update in EM Conference Highlights 2015
For Salim Rezaie’s Best Case Ever on PE see Best Case Ever 44 Low Risk Pulmonary Embolism
Update 2016 CHEST Guidelines on management of pulmonary embolism
Dr. Helman, Dr. Chopra and Dr. Foote have no conflicts of interest to declare
Key References
Aujesky et al. Lancet. 2011;378: 21. www.ncbi.nlm.nih.gov/pubmed/21703676
Douma et al. BMJ. 2010; 340: c1475. www.ncbi.nlm.nih.gov/pubmed/20354012
Jaff et al. Circulation. 2011; 123; 1788. www.ncbi.nlm.nih.gov/pubmed/21422387
Leung et al.Am J Respir Crit Care. 2010;184:1200. www.ncbi.nlm.nih.gov/pubmed/22086989
Rizkallah et al. Chest. 2009;135:786. www.ncbi.nlm.nih.gov/pubmed/18812453
Tillie-Leblond et al.Ann Intern Med. 2006;144:390. www.ncbi.nlm.nih.gov/pubmed/16549851
Wells et al.Thromb Haemos. 2000; 83:416. www.ncbi.nlm.nih.gov/pubmed/10744147
Witting MD, Mattu A, Rogers R, Halvorson C. Simultaneous T-wave inversions in anterior and inferior leads: an uncommon sign of pulmonary embolism. J Emerg Med. 2012;43(2):228-35. http://www.ncbi.nlm.nih.gov/pubmed/22142671
For more on Pulmonary Embolism on EM Cases:
Best Case Ever 44 Low Risk Pulmonary Embolism
Ep 113 Pulmonary Embolism Challenges in Diagnosis Part 1
Ep 114 Pulmonary Embolism Challenges in Diagnosis 2 – Imaging, Pregnancy, Subsegmental PE
More FOAMed Resources on Pulmonary Embolism
Scott Weingart discusses Pulmonary Embolism Treatment Options on EMCrit
Brit Long blogs on Pulmonary Embolism: Management of the Unstable Patient on emDocs






Leave A Comment