In this month’s main episode podcast on Urologic Emergencies – Priapism and Urinary Retention with Dr. Natalie Wolpert and Dr. Yonah Krakowsky we answer questions such as: for priapism, how much time to do we have to fix it before there’s irreversible tissue damage? How is priapism managed differently depending on the cause? What is the value of a corporal blood gas for managing priapism? What are the indications for cavernosal phenylephrine injections? What are the common medications that cause urinary retention that we often miss leading to needless recurrent urinary retention? Why is a suprapubic catheter in many respects safer than a urethral catheter for managing urinary retention? Which patients are at high risk for complications of post-obstructive diuresis? and many more…
Podcast production, sound design & editing by Anton Helman
Written Summary and blog post by Shaun Mehta and Deb Saswata, edited by Anton Helman July, 2020
Cite this podcast as: Helman, A. Episode 143 Priapism and Urinary Retention: Diagnosis and Management. Emergency Medicine Cases. July, 2020. https://emergencymedicinecases.com/priapism-urinary-retention. Accessed [date]
Priapism: The Ischemic Fifth Limb
Priapism is defined as an unwanted prolonged erection. Similar to acute brain and heart ischemia, time is tissue for ischemic priapism. Initiate treatment as soon as possible, preferably within 4-6 hours to minimize the risk of impotence that occurs in 100% of patients with untreated ischemic priapism at 48 hours.
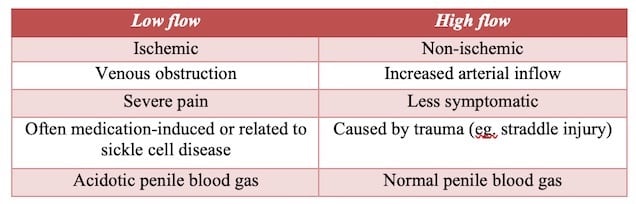
Low flow vs. High flow Priapism
Low flow priapism is ischemic and a true urologic emergency – a compartment syndrome of the penis, whereas high flow is non-ischemic. Low flow is far more common, with high flow only making up about 2% of presentations.
Priapism is a clinical diagnosis. On exam, key findings include an erect corpus cavernosa with a flaccid glans. There are a number of ways to differentiate ischemic and non-ischemic priapism based on history and lab findings.
A variety of medications and toxins can cause ischemic priapism, including:
- Intra-cavernosal injections (“triple mix” – papaverine, prostaglandin E1 and phentolamine)
- PDE5 inhibitors (sildenafil, tadalafil)
- Anti-hypertensives (hydralazine, prazosin, calcium channel blockers)
- Neuroleptics (trazodone, chlorpromazine)
- Drugs of abuse: cocaine, marijuana
Get a baseline penile blood gas with the first aspiration of intra-cavernosal blood. Although it may not aid in the diagnosis, serial gases may be useful to monitor response to treatment. An ischemic blood gas will be dark, hypoxemic (pO2<30-40), acidotic (pH <7.25, pCO2 >60) and glucopenic.
Management of priapism: stepwise approach
- Dorsal nerve block: retract the penis caudally and insert a small gauge (25-27G) needle on either side of the midline at 10 and 2 o’clock, inject lidocaine (without epinephrine); you should feel a pop when you pass through Buck’s fascia to know you’re in the correct space Dorsal nerve block video here
- Corporal aspiration (getting blood out of the penis): insert a 19G butterfly needle into the lateral corpora at the 10 and 2 o’clock positions; aspirate 10-20 mL of blood (while the patient is squeezing the penis proximally) and send a blood gas; avoid the urethra (ventrally) and neurovascular bundle (dorsally); this can be repeated on the other side if priapism persists; a patient’s response to this treatment largely depends on how long they have had an erection
- Corporal irrigation: if detumescence does not occur after 2 aspiration attempts of 20-25 mL each, irrigate the corpus cavernosa with 25 mL of cold (10°C) sterile saline; aspirate the fluid back after a period of 20 minutes if priapism persists
- Phenylephrine injection squeezes the blood out of the penis and back into the body; ask the patient to squeeze the penis distally to help facilitate this, dilute to 100 mcg/mL and inject 1-2 mL q5min, to a maximum dose = 1 mg over 1 hour.
- Consult urology for consideration of surgical management
Clinical Pearl: for aspiration of the corpora, ask the patient to squeeze their penis proximally to get the blood out of the body; for phenylephrine or cold saline injections ask the patient to squeeze their penis distally to get the blood from the penis into their body.
Other ischemic priapism management options
- Terbutaline 5-10 mg orally has some supporting evidence
- Exercise – ask them to do squats or walk up and down a few flights of stairs; there are case reports claiming effectiveness
Urinary Retention
While the diagnosis of urinary retention is usually straight forward, an important first step or cognitive forcing strategy is to consider the life- or limb-threatening etiologies such as cauda equina syndrome/spinal cord compression.
Causes of urinary retention
The 4 main categories that can cause urinary retention are obstructive, infectious/inflammatory, neurologic, and medications.
Clinical Pearl: While obstructive pathology such as an enlarged prostate may be a common etiology of urinary retention, a neurologic cause such as cauda equina syndrome or cord compression (for example, secondary to a metastatic spinal mass from prostate cancer) should be considered and ruled out when assessing patients for retention.
Indications for a urethral catheter for patients in urinary retention
The most common indication for insertion of a urethral catheter is in the patient who is unable urinate (or is dribbling urine) for 10-12 hours with significant lower abdominal discomfort and bladder distension. Other indications for urethral catheter insertion include acute kidney injury (elevated creatinine or hydronephrosis) secondary to an obstruction, a patient with a suspected urinary tract infection who is unable to void, or evidence of overflow incontinence.
A post-void residual bladder scan may be helpful in confirming urinary retention. There is variation in defining urinary retention according to post-void residual urine volume ranging from 100-300 mL. However, volumes <100 mL are generally considered a rule out while volumes >300 mL are generally considered a rule in.
Approach to difficult urinary catheter insertion: Key steps
Analgesia: consider a short acting systemic analgesic such as fentanyl. Apply ample amounts of lubricant with lidocaine (e.g. 2 uro-jets). It is important that the patient is as relaxed as possible so that the pelvic muscles become relaxed.
Clinical Pitfall: injecting the lubricant with lidocaine into the meatus too quickly can be painful and may cause the patient to tighten/tense the pelvic muscles thus preventing passing of the urethral catheter into the bladder. Intraurethral injection must be done slowly.
Choice of urethral catheter: the most common catheter used in the setting of a difficult urethral catheter insertion is a 16-F Coude catheter.

Image source from prome supply
Clinical Pearl: if the cause of retention is not a stricture, but rather an enlarged prostate, the size of the catheter should be increased from 16-F to 18-F if the insertion of the 16-F was unsuccessful. A thicker catheter is less likely to find the many crevices present in an enlarged prostate. In contrast, if the cause of the retention is a stricture, a smaller catheter size should be chosen.
Technique: the penis should be held perpendicular to the patient (the meatus should be facing the ceiling with the patient supine). Inject ample amounts of lubricant with lidocaine slowly and gently pinch the meatus after injection to prevent the lubricant from leaking back outside of the penis.
Clinical Pearl: if the patient has a history of radical prostatectomy and if the Coude catheter is not passing, think bladder neck obstruction and consult urology.
Indications for suprapubic catheter
Suprapubic catheters are considered after unsuccessful attempts with urethral catheters usually in the setting of severe urethral strictures or complex prostatic disease. Another indication for suprapubic catheter is urethral disruption due to trauma. While indwelling urethral foley catheters remain the initial approach to urinary retention, suprapubic catheters have been shown to be more comfortable, associated with less bacteriuria and less re-catheterization rates compared to indwelling urethral catheters.
Contraindications to placing a suprapubic catheter include empty or unidentifiable bladder and bowel anterior to the bladder wall.
Suprapubic catheter key procedural steps
Video suprapubic catheter insertion
-Gear required includes peel-away introducer sheath (can use central line kit using the Seldinger technique if not available), ultrasound, sterile gloves, chlorhexidine, lidocaine, sterile syringe, spinal needle (22 gauge for adults), scalpel, foley catheter, dressing
-Ultrasound to identify and landmark the bladder (usually 1-2 cm above pubis symphysis)
-Prep skin and local anaesthetic
-Using real-time ultrasound guidance, advance needle to bladder
-Placement is confirmed when urine is withdrawn into syringe (can also confirm on ultrasound)
-As a temporizing measure (when, for example there is poor access to a urologist and you have little/no experience advancing a wire into the bladder), much of the urine can be withdrawn at this step to relieve the bladder distention and the procedure terminated until help arrives
-Advance guidewire into bladder
-Use scalpel to extend skin incision to allow for dilator
-Pass peel-away sheath and dilator over guidewire
-Insert foley catheter through sheath and into bladder
-Aspirate urine to confirm foley placement and inflate foley balloon
-Remove peel-away sheath and dress site
Post-obstructive diuresis: when to worry
Post-obstructive diuresis is defined as urinary output > 200 mL for at least 2 hours after urethral catheter insertion, or > 3L in 24hrs. This is after the initial volume of urine has come out.
Patients who are at high risk of complications as a result of post-obstructive diuresis include patients with abnormal electrolytes or newly elevated creatinine, volume overload, uremic, or confused. These patients should be observed for at least 4 hours following urethral catheter insertion and if urinary output is > 200ml/hr, they should be admitted with a consult to internal medicine.
Low risk patients (normal electrolytes/creatinine, euvolemic, clinically well) usually do not require observation after the urethral catheter is inserted and distended bladder is relieved.
Duration of indwelling urethral catheter
Our experts recommend that urethral indwelling catheters remain in the bladder for a duration of 7-10 days. If the catheter is removed too early (ie. in 2-3 days), the likelihood of requiring re-catheterization (with potential complications of repeated catheterization) has shown to be high. Antibiotic prophylaxis is not recommended unless there is an underlying infection present. In patients with benign prostatic hypertrophy as a cause of retention, an alpha-blocker such as tamsulosin 0.4mg daily has been shown to decrease the likelihood of re-catheterization after a trial of void. Exercise caution in prescribing alpha-blockers in patients with Parkinson’s disease and/or orthostatic hypotension. All patients should be educated regarding orthostatic hypotension when prescribing alpha blockers.
Common clinical pitfall: a common pitfall is to remove the urethral catheter after 2-3 days when patients request to do so; the evidence is clear that the likelihood of re-catheterization and it’s associated traumatic complications are higher compared to removal after 7 days.
Take Home Points for priapism and urinary retention
- Priapism: time is tissue; treatment should be started by the emergency physician, not the urologist
- Assume ischemic priapism until proven otherwise
- In urinary retention, important to consider and rule out the dangerous neurologic causes including cauda equina syndrome
- Use lots of lubricant with lidocaine for urethral catheter insertion and inject slowly
- In the setting of enlarged prostate, if a 16-F Coude catheter insertion is unsuccessful, then increase the size of the catheter
- Patients with high risk of post-obstructive diuresis and it’s complications should be observed in the ED for at least 4 hours
- The urethral catheter should remain in-situ for at least 1 week and an alpha-blocker should be considered to decrease the likelihood of re-catheterization after a trial of void
References
Priapism
- Montague DK, Jarow J, Broderick GA, et al. American Urological Association guideline on the management of pria- pism. J Urol. 2003;170(4 Pt 1):1318-1324.
- Salonia A, Eardley I, Giuliano F, et al. Europe an Association of Urology guidelines on priapism. Eur Urol. 2014;65(2):480- 489.
- Ateyah A, Rahman El-Nashar A, Zohdy W, et al. Intracavernosal irrigation by cold saline as a simple method of treating iatrogenic prolonged erection. J Sex Med. 2005;2(2):248-253.
- Lander J, Brady-Fryer B, Metcalfe JB, et al. Comparison of ring block, dorsal penile nerve block, and topical anesthesia for neonatal circumcision: a randomized controlled trial. JAMA. 1997;278(24):2157-2162.
- Priyadarshi S. Oral terbutaline in the management of pharmacologically induced prolonged erection. Int J Impot Res. 2004;16:424-426.
- Chinegwundoh FI, Smith S, Anie KA. Treatments for priapism in boys and men with sickle cell disease. Cochrane Database Syst Rev. 2017;9:CD004198.
- Lowe FC, Jarow JP. Placebo-controlled study of oral terbutaline and pseudoephedrine in management of prostaglandin E1-induced prolonged erections. Urology. Jul 1993;42(1):51-3
- Priyadarshi S. Oral terbutaline in the management of pharmacologically induced prolonged erection. Int J Impot Res 2004;16(5):424–6.
- Kazuyoshi, S. Namiki, M. Clinical Management of Priapism: A Review. Word J Mens Health. 2016 April; 34(1): 1-8.
- Habous, M et al. Noninvasive treatments for iatrogenic priapism: Do they really work? A prospective multicenter study. Urol Ann. 2016 Apr-Jun; 8(2): 193-196.
- Priyadarshi S. Oral terbutaline in the management of pharmacologically induced prolonged erection. Int J Impot Res 2004;16(5):424-6.
- Gravel J, Leblanc C, Varner C. Management of priapism with a trial of exercise in the emergency department. CJEM. 2019;21(1):150-153.
Urinary Retention
- Karavitakis M, Kyriazis I, Omar MI, et al. Management of Urinary Retention in Patients with Benign Prostatic Obstruction: A Systematic Review and Meta-analysis. Eur Urol. 2019;75(5):788-798.
- Fisher E, Subramonian K, Omar MI. The role of alpha blockers prior to removal of urethral catheter for acute urinary retention in men. Cochrane Database Syst Rev. 2014;(6):CD006744.
- Hassan S, El-Ebiary M, Mabrouk M. Early versus late trail of catheter removal in patients with urinary retention secondary to benign prostatic hyperplasia under tamsulosin treatment. Urol Sci 2018;29:288–92.
- Verhamme KM, Dieleman JP, Van Wijk MA, et al. Nonsteroidal anti- inflammatory drugs and increased risk of acute urinary retention. Arch Intern Med. 2005;165(13):1547-1551.
- Nyman MA, Schwenk NM, Silverstein MD. Management of urinary retention: rapid versus gradual decompression and risk of complica- tions. Mayo Clin Proc. 1997;72(10):951-956.
- Brosnahan J, Jull A, Tracy C. Types of urethral catheters for manage- ment of short-term voiding problems in hospitalised adults. Cochrane Database Syst Rev. 2004;(1):CD004013.
- Boettcher S, Brandt AS, Roth S, et al. Urinary retention: benefit of gradual bladder decompression – myth or truth? A randomized controlled trial. Urol Int 2013;91(2): 140–4.
- Niel-Weise BS, van den Broek PJ. Urinary catheter policies for short- term bladder drainage in adults. Cochrane Database Syst Rev. 2005;(3): CD004203.
- McPhail MJ, Abu-Hilal M, Johnson CD. A meta-analysis comparing suprapubic and transurethral catheterization for bladder drainage after abdominal surgery. Br J Surg. 2006;93(9):1038-1044.
- Desgrandchamps F, De La Taille A, Doublet JD, RetenFrance Study Group. The management of acute urinary retention in France: a cross-sectional survey in 2618 men with benign prostatic hyperplasia. BJU Int 2006;97:727.
- Djavan B, Shariat S, Omar M, et al. Does prolonged catheter drainage improve the chance of recovering voluntary voiding after acute urinary retention (AUR)? Eur Urol 1998;33(Suppl. 1):110.
Other FOAMed resources on pripism and urinary retention
- https://coreem.net/core/priapism/
- http://www.emdocs.net/priapism-ed-pearls-pitfalls/
- https://www.nuemblog.com/blog/priapism
Drs. Helman, Wolpert and Krakaowsky have no conflicts of interest to declare
Now test your knowledge with a quiz.








Why lidocaine without epinephrine for the penile block? If sympathomimetics are being given intracorporally and the point is to clamp down on arterial flow, I don’t see the harm in using lido w/epi.
Great lecture! I have always been in the habit of sending everyone home with a foley still in — but the question came up reccently about a man who had retention after anesthesia earlier that day. He did have a history of BPH. He was not excited about the foley staying in and it made me wonder if, in that situation, we can send him home without one with good return precautions. After anesthesia, what are the chances of the retention recurring if we leave the catheter out?
This podcast was an absolute home run. I almost want a priapism to come in now. You all demystified the management for me like no one else could after 20 years of medicine. I would reiterate the aggressiveness of management really depends on the clinical setting one is in – with easy or difficult urology access the management really feels different. Thank you!
Would sublingual nitro help relieve priapism by vein vasodilatation? Was it ever tried or studied?
Amazing work again, thank you for all the knowledge!