What’s in the name – why “Massive Hemorrhage Protocol” and not “Massive Transfusion Protocol”? In this episode – The 7 Ts of Massive Hemorrhage Protocols, Dr. Jeannie Callum, Dr. Andrew Petrosoniak and Dr. Barbara Haas join Anton in answering the questions: How do you decide when to activate the MHP? How do you know when it is safe to terminate the MHP? What lab tests need to be done, how often, and how should the results be shared with the clinical team? Once the dust settles, what do we need to tell the patient and/or their family about the consequences of being massively transfused? What should be the lab resuscitation targets? Why is serum calcium important to draw in the ED for the patient who is exsanguinating? How do we mitigate the risk of hypothermia? What can hospitals do to mitigate blood wastage? If someone is on anti-platelets or anticoagulants what is the best strategy to ensure the docs in the ED know what to give and how much? Until the results of lab testing come back and hemorrhage pace is slowed, what ratio of plasma to RBCs should we target? What’s better, 1:1:1 or 2:1:1? Should we ever consider using Recombinant Factor 7a? If the fibrinogen is low, what is the optimal product and threshold for replacement? When and how much TXA? Anyone you wouldn’t give it to? and many more…
Podcast production, sound design & editing by Anton Helman;
Written Summary and blog post by Saswata Deb & Priyank Bhatnagar, edited by Anton Helman;
Infographic by Pouria Rezapour, edited by Anton Helman, February, 2021
Cite this podcast as: Helman, A. Callum, J. Haas, B. Petrosoniak, A. The 7 Ts of Massive Hemorrhage Protocols. Emergency Medicine Cases. February, 2021. https://emergencymedicinecases.com/7-ts-massive-hemorrhage-protocols. Accessed [date]
When a patient is exsanguinating, having a standardized massive hemorrhage protocol (MHP) enables rapid and coordinated delivery of life-saving blood products, medications. Every one-minute delay in receiving the first pack red blood cells (pRBC) in a bleeding trauma patient is associated with a 5% increase in mortality.
Why Massive Hemorrhage Protocol instead of Massive Transfusion Protocol?
The classic definition of massive transfusion protocol is 10 units of pRBC over 24 hours and only focuses on the number of blood products transfused. Instead, the emphasis should be placed on hemorrhage control which not only includes transfusions, but also monitoring blood work/considering targets, administration of other medications including tranexamic acid, keeping the patient warm and source control of the bleeding.
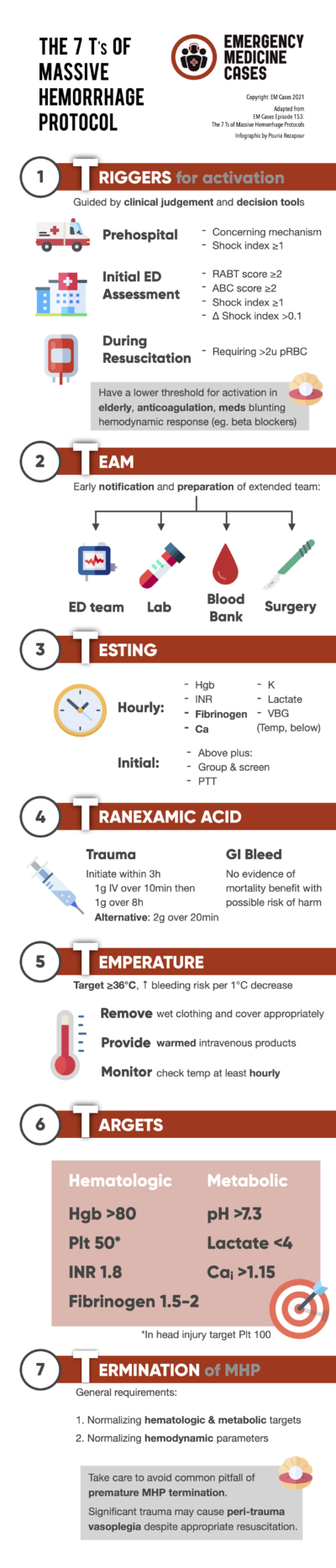
The 7 Ts of Massive Hemorrhage Protocols: Trigger, Team, Testing, TXA, Temperature, Target, Termination
Trigger: indications for MHP prehospital, at initial ED assessment and during resuscitation
The decision to active a MHP should be guided by clinical judgement, decision tools and response to treatment.
A) Prehospital indications for MHP include a concerning mechanism of injury (eg., fall from ≥ 3 stories) and/or a shock index ≥ 1.
B) Initial ED assessment indications for MHP include clinical judgement incorporating clinical decision tools, resuscitation intensity and pitfall conditions.
- Clinical judgement (obvious shock state or else incorporating shock index >1, delta shock index ≥0.1, RABT Score ≥2, AND
- Pitfall conditions – consider a lower threshold for activating MHP
-
- Older
- On anticoagulants or dual anti-platelet therapy – patients taking anticoagulants are not well represented in MHP studies; have a lower threshold for calling for blood
- Medications that may blunt hemodynamic parameters despite bleeding out (ie., beta-blockers)
C) Indications for MHP during resuscitation include resuscitation intensity (see below) or requiring >2u pRBCs
The Revised Assessment of Bleeding and Transfusion (RABT) Score – 1 point each
- Shock index > 1.0
- Pelvic Fracture
- Positive FAST
- Penetrating Injury
A 2018 study showed that RABT score ≥ 2 performed better than ABC score in predicting need for MHP.
Resuscitation intensity to help guide the decision to active a MHP
If hemorrhagic shock is not obvious clinically, consider the resuscitation intensity: patients who require 3 units of any combination of crystalloids or blood products to maintain adequate perfusion are considered to have high resuscitation intensity which predicts higher mortality, and should be considered for activation of the MHP.
2:1:1 or 1:1:1 blood product ratio?
Our experts recommend a 2:1:1 ratio of blood products based on their interpretation of the PROPPR trial which found that among patients with severe trauma and major bleeding there was no significant difference in 24hr or 30 day mortality in patients who received a 1:1:1 ratio compared to a 2:1:1 ratio, and because of practical considerations that allow faster administration of blood products using the 2:1:1 ratio. They therefore recommend:
- The first case of blood products should contain 4 units uncrossmatched pRBCs, be at the bedside in under 10 minutes, and IV running via rapid transfuser shortly thereafter
- Then the next case of blood products contains 4 RBC and 4 plasma (FFP) which should run simultaneously
“European” Fibrinogen +/- PCCs up front vs “North American” plasma strategy
Using fibrinogen concentrates with or without PCCs up front in the hemorrhaging trauma patient is common in many European countries. The small randomized studies suggest that this strategy is at least as good as the classic North American plasma strategy at 1:1. There are some benefits to the concentrate strategy – you can keep these products at room temperature in a near patient location, a blood group is not necessary, it avoids the need for AB plasma that is often in short supply, they are pathogen-reduced, small volumes, injected over minutes, and firbrinogen/PCCs do not cause transfusion related acute lung injury TRALI.
Optimal threshold for replacement of fibrinogen?
There are only a few small poorly controlled studies that compare low vs. high threshold of fibrinogen level for controlling blood loss. The studies do suggest higher might be better. Guidelines recommend a fibrinogen threshold for transfusion of fibrinogen or cryoprecipitate of <1.5-2.0 g/L.
Team: prepare your gear, yourself and your team
Early notification and preparation of the extended team, including the ED team, laboratory team, blood bank and surgical team is essential to ensure rapid delivery of blood products and bleeding source control. Based on the limited data you gather from EMS, brief the ED team, prioritizing objectives (e.g. may require tourniquet before securing airway, priority to administer blood products) and adapt a shared mental model. Team members should have roles assigned including which gear they each need to get to the bedside before the patient arrives, if time permits.
Testing: initial baseline tests and repeated testing
Having a standardized order set for laboratory investigations in MHP can help reducing cognitive burden and prevent missed diagnoses of coagulopathy or metabolic dysfunction in hemorrhage.
Initial baseline tests: CBC, coags (INR, PTT, fibrinogen), electrolytes (including calcium), VBG, lactate +/- BhCG
Q1h tests: Hb, INR, lactate, VBG, fibrinogen
Calcium plays an important role in regulating coagulation and hemostasis. The citrate preservative in blood products binds to serum calcium making it inactive. It is thus vital to monitor serum calcium and to consider administering calcium every 3 to 4 blood products that are administered.
Fibrinogen is often forgotten in lab orders and is impacted by both coagulopathy of trauma and dilution from administered blood products. It requires careful monitoring and replacement with cryoprecipitate or fibrinogen concentrate.
A high INR has been shown to predict poor outcomes for massively hemorrhaging patients and correcting it with FPP +/- PCCs (see below) is vital. It should also be used to assess the trajectory of the resuscitation and should be checked every hour or every 3 to 4 units of pRBCs along with Hb, fibrinogen and lactate.
PTT is a one time screening test – no need for repeating. A single measurement of PTT early in the resuscitation helps screen for congenital bleeding disorders or on-board anticoagulants, such as DOACs, that may need to be co-managed.
Pitfall: Calcium and fibrinogen are the blood tests that are most often forgotten in severely hemorrhaging patients; use a checklist/order set that includes these to avoid this pitfall
SAMPLE MASSIVE HEMORRHAGE PROTOCOL CHECKLIST (for educational purposes only)
Tranexamic Acid (TXA) indications, contraindications and dosing
The decision to give TXA should be made at the same time as the decision to give blood products in the hemorrhaging trauma patient – early!
Our experts recommend TXA for all trauma patients in whom you suspect life-threatening hemorrhage within 3 hours of the time of injury who are receiving blood products for hemorrhaging, and in patients with initial SBP<90 or HR >110 based on the CRASH-2 trial. Give TXA ASAP – observational data suggests that every 15 min delay decreases its mortality benefit by 10%.
For isolated head injured patients the CRASH-3 trial did not show a clinically significant benefit for early administration of TXA.
Relative contraindications to TXA
- History of coronary stent(s)
- Active hematuria (it is thought that administration of TXA in the patient with hematuria may cause clot formation resulting in obstructive uropathy)
- History of venous thromboembolic disease
Pitfall: TXA is sometimes forgotten; if you are transferring a patient to a trauma center with concern for active hemorrhage, be sure to administer TXA prior to transport within 3 hours of the time of injury (the earlier the better).
TXA dosing
Observational data suggests that about 50% of the time the second bolus or infusion of TXA after the initial 1g bolus is not given at all in trauma patients, likely because the infusion is associated with logistical problems of requiring a dedicated IV. Some patients will not be adequately reversed with 1g alone. Our experts recommend a one-time 2g IV dose up front to ensure all patients receive an adequate dose of TXA.
Learn more about indications for TXA at Journal Jam 18: The Evidence of TXA
Temperature: hypothermia increases mortality in trauma patients
Hypothermia, which results from the trauma itself as well as the administration of blood products, is sometimes overlooked in the management of polytrauma patients. Hypothermic patients have poor outcomes because of the negative effects of hypothermia on coagulation. It is important to employ early temperature measurement in a systematic way by incorporating it into a trauma checklist and repeating the measurement q1h, or more frequently if the patient was initially hypothermic. Simple methods to keep the patient warm include removing wet clothing, placing warm blankets, administering warmed IV products and monitoring temperature q1h and more often in patients who present hypothermic.
Targets: clinical, hematologic and metabolic resuscitation targets
There are clinical and laboratory resuscitation targets that should be used to help monitor patient stability, response to resuscitation efforts and for prognostication. Laboratory targets are more important to assess trajectory of MHP and other resuscitation efforts rather than initial assessment, and can be divided into hematologic and metabolic.
Clinical targets to consider include HR <100, MAP >55-70 (depending on baseline BP) GCS >15 (if no head injury/intoxication), urine output >30mL/hr and normal IVC diameter/collapsibility

Hematologic and metabolic targets in trauma patients in whom a massive hemorrhage protocol has been activated
Termination of MHP
The decision to stop a MHP is nuanced and based on hemodynamics and resuscitation targets outlined above. It is important to involve both the ED and inpatient teams in the decision to terminate MHP to avoid a second wave of bleeding and instability. Ideally, MHP termination conversations should happen when both patient hemodynamics and resuscitation targets are normalized or at least trending in the right direction.
General requirements for termination of MHP include
- Normalizing hematologic and metabolic targets
- Normalizing hemodynamic parameters
Pitfall: take care to avoid the common pitfall of premature MHP termination; significant trauma may cause peri-trauma vasoplegia despite appropriate resuscitation
Special Considerations in Massive Hemorrhage Protocols
Smaller hospitals with challenging access to blood products
In rural/remote hospital settings where blood products may not be available, the use of prothrombin concentrate complex (PCCs) and fibrinogen should be considered. In most European centres, red cells along with fibrinogen with or without PCC (instead of FFP and platelets) have become common practice (see above). While there are multiple benefits of the concentrate strategy (can be stored at room temperature, not requiring a blood group, pathogen-reduced, small volumes, injected over minutes, and avoids TRALI reactions), the benefits have only been shown in small studies. Large trials are required to further corroborate these findings.
Life threatening hemorrhage on warfarin
If the patient is known to be taking warfarin and has imminent life-threatening bleeding and a MHP is being activated, our experts recommend both FFP at a minimum ratio of 1:2 and 2000 International Units (IU) PCC, along with vitamin K 10mg to ensure sustained reversal in 6 hours after the PCCs wear off. It is not necessary to wait for the INR result before administering PCCs in this context.
Pearl: some patients who have a very high INR and/or morbid obesity, or have continued significant bleeding 1hr after the first dose may require a 2nd dose of PCC 2000 IU 1hr later, preferably in consultation with a hematologist
Life threatening hemorrhage on Direct Oral Anticoagulants (DOACs)
While the evidence is weak with respect to clinical benefit, an acceptable regimen in the trauma patient with life threatening bleeding who has taken a DOAC within 12 hours, in whom a MHP has been activated is:
- For a patient on Dabigatran consider Idarucizumab 5g
- For a patient on a Xa inhibitor (eg., apixaban, rivaroxaban), PCC 2000 IU; if significant bleeding persists after 1 hour, a second dose of 2000 IU of PCC should be considered; while not approved in Canada, a specific antidote to Xa inhibitors, Andexanet alfa, has also been used in these situations as a continuous infusion.
Life threatening hemorrhage in obstetrical patients
Observational data has shown that a fibrinogen level below 2g/L leads to worse outcomes including maternal death, in the bleeding obstetrical patient. Our experts recommend IV fibrinogen concentrate 4g or cryoprecipitate 10 units, as soon as possible and then check the fibrinogen level to ensure it is >2g/L. Replacement of fibrinogen may decrease both the need for ongoing blood transfusions and associated complications such as volume overload.
Pearl: pregnant and postpartum patients who are hemorrhaging tend to have low fibrinogen with an increased risk for DIC, so have a low threshold to give fibrinogen in the massively hemorrhaging obstetrical patient.
Beware: Blood transfusions are not benign – consider them like an organ transplant; female patients of childbearing age should receive O-negative pRBCs until crossmatch is obtained; they should also be counselled to have a group and screen completed 3 to 6 months post-transfusion due to associated alloimmunization risks.
Blood product stewardship: practical methods to prevent wastage
Blood products are a limited resource in hospitals that require careful utilization in appropriate clinical indications. Methods to prevent blood product wastage are:
- Ensure indication for MHP is met; for example, a patient who requires transfusion of only 2 units of red cells to reach clinical and lab targets does not require ongoing MHP which includes FFP and platelets
- Return unused blood products in the same package configuration within the container as they came from the blood bank to prevent damage from improper storage
- Unused blood products must be returned within 60 minutes to prevent wastage
- Do not write on the blood product package labels as this would render the product unusable if returned
- Do not return empty blood product packages with unused products as this produces risk of contamination and safety hazards for blood bank technicians
Take Home Points for the 7 Ts of Massive Hemorrhage Protocols
- Have a formalized MHP in your institution and between institutions so that bleeding patients receive state-of-the-art standardized care irrespective of which institution they are in
- Time is life! Every one-minute delay in receiving the first pRBC is associated with a 5% increase in mortality
- Trigger for MHP should take into account clinical judgement based on obvious shock state, shock index >1, delta shock index ≥0.1, RABT Score ≥2, resuscitation intensity), mechanism of injury and pitfall conditions (older, anticoagulants, certain drugs that alter vital signs)
- Both FFP and PCC along with vitamin K are sometimes required to reverse INR in a patient on warfarin with life-threatening hemorrhage, and repeat dosing of PCC in 1hr should be considered for patients with very high INR, morbidly obese patients and those with significant continued bleeding
- Fibrinogen is the first coagulation factor to drop in trauma and needs to be replaced and measured to assess trajectory
- Pregnant and postpartum patients who are hemorrhaging tend to have low fibrinogen with an increased risk for DIC, so have a low threshold to give fibrinogen in the massively hemorrhaging obstetrical patient
- Patients in whom a MHP is activated tend to develop hypocalcemia; it is important to monitor serum calcium levels and replace calcium accordingly
- Do not neglect to keep the patient warm by monitoring temperature, removing wet clothing, using warm blankets and warmed IV products
- TXA administered within 3 hours of injury has a significant mortality benefit – be sure to give it prior to transfer to a trauma center (the earlier the better)
- Termination of MHP is a shared decision amongst the ED and inpatient teams and should take into account both clinical and laboratory targets, taking care not to prematurely terminate the MHP (which can lead to a second wave of hemorrhage and instability)
- There are easy ways to prevent blood product wastage so that those blood products can be used to save more lives
SAMPLE MASSIVE HEMORRHAGE PROTOCOL CHECKLIST (for educational purposes only)
References
-
- Callum, J et al. A regional massive hemorrhage protocol developed through a modified Delphi technique. CMAJ. September 3, 2019 vol. 7 no. 3 E546-E561.
- Vandromme MJ, Griffin RL, Kerby JD, et al. Identifying risk for massive transfusion in the relatively normotensive patient: utility of the prehospital shock index. J Trauma 2011;70(2):384–8 [discussion: 388–90].
- Schellenberg M, Strumwasser A, Grabo D, et al. Delta Shock Index in the Emergency Department Predicts Mortality and Need for Blood Transfusion in Trauma Patients. AmSurg. 2017;83(10):1059-1062.
- Joseph B et al. Massive Transfusion: The Revised Assessment of Bleeding and Transfusion (RABT) Score. World J Surg 2018 Nov 42(11): 3560-3567.
- Rahbar E, Fox EE, Del junco DJ, et al. Early resuscitation intensity as a surrogate for bleeding severity and early mortality in the PROMMTT study. J Trauma Acute Care Surg. 2013;75(1 Suppl 1):S16-23.
- Meyer DE, Cotton BA, Fox EE, et al. A comparison of resuscitation intensity and critical administration threshold in predicting early mortality among bleeding patients: A multicenter validation in 680 major transfusion patients. J Trauma Acute Care Surg. 2018;85(4):691-696.
- Holcomb JB, Tilley BC, Baraniuk S, et al. Transfusion of Plasma, Platelets, and Red Blood Cells in a 1:1:1 vs a 1:1:2 Ratio and Mortality in Patients With Severe Trauma JAMA. 2015; 313(5):471-478.
- Levy, J. H., Welsby, I., & Goodnough, L. T. (2013). Fibrinogen as a therapeutic target for bleeding: A review of critical levels and replacement therapy. Transfusion, 54(5), 1389-1405.
- Sahin, A. S., & Ozkan, S. (2019). Treatment of obstetric hemorrhage with fibrinogen concentrate. Medical Science Monitor, 25, 1814-1821.
- Kander, T., & Schött, U. (2019). Effect of hypothermia on haemostasis and bleeding risk: A narrative review. Journal of International Medical Research, 47(8), 3559-3568.
- Effects of tranexamic acid on death, vascular occlusive events, and blood transfusion in trauma patients with significant haemorrhage (CRASH-2): A randomised, placebo-controlled trial. (2010). The Lancet, 376(9734), 23-32.
FOAMed Resources on Massive Hemorrhage Protocols
- Massive Transfusion Protocol- EM Crit
- Broome Docs Massive Transfusion Protocol for rural docs
- Transfusions, Anticoagulants and Bleeding on EM Cases
- First10EM analysis of CRASH-2 trial
- REBELEM analysis of PROPPR trial
- Andrew Petrosoniak’s talk at EMU on “Bleeding Out: Massive Transfusion Protocols”
- EM Quick Hit on TXA in GI bleeds
- emDOCs Massive Blood Transfusion
- Logistics of administering massive transfusion on EM Crit
This podcast was supported by a Canadian Institutes of Health Research grant for a study looking at transfusion products and ratios in trauma patients
Now test your knowledge with a quiz.





Thanks so much for this amazing lecture
This is my first point of learning about MHP. Thank you for such a concise and descriptive overview and expert opinion.
Excellent presentation and most content are covered so well and easy to follow instructions to all Ed physicians.
Thank you.
With the 2:1:1 ratio Is it within the first 24 hours or simultaneous as the resuscitation is occurring?
24 hours. Practically speaking in the ED we suggest getting 4 units red cells to bedside within 10 mins and start them via rapid transfuser. The 2nd container of blood products should contain 4 units of red cells and 4 units of plasma that should run simultaneously.