This is EM Cases Episode 88 – DOACs Part 1: Use and Misuse, DVTs and atrial fibrillation.
As we get better at picking up thromboembolic disease, and the indications for Direct Oral Anticoagulants (DOACs) widen, we’re faced with increasingly complex decisions about when to start these medications, how to start them, when to stop them and how to manage bleeding associated with them. There’s a lot that we need to know about these drugs to minimize the risk of thromboembolism in our patients while at the same time minimizing their risk of bleeding.
So with these goals in mind, Dr. Walter Himmel – the walking encyclopedia of EM, Dr. Jim Douketis, one of the world’s most published researchers in thrombosis and Dr. Ben Bell, internist extraordinaire will answer such questions as: What are the important patient factors to take into account before starting a DOAC? What baseline blood work is required? How do DOACs work? How does their bleeding risk compare to Warfarin? Are there any practical blood tests to help monitor these drugs? Which patients with isolated calf DVTs require anticoagulation? Which patients with superficial venous thrombosis require anticoagulation? Which patients taking antiplatelet agents who you’re starting on a DOAC should be kept on the antiplatelet agent? Which patients with atrial fibrillation should be started on a DOAC in the ED? and many more…
Written Summary and blog post by Anton Helman, edited by Benjamin Bell, November, 2016
Cite this podcast as: Helman, A, Himmel, W, Douketis, J, Bell, B. DOACs Part 1: Use and Misuse. Emergency Medicine Cases. https://emergencymedicinecases.com/doacs-use-misuse/. Accessed [date].
Important patient factors to take into account before considering DOACs
The following are contraindications, relative contraindications or patient populations in which the evidence for DOACs’ effectiveness and safety has not been established.
Older age: In patients >80 years old with atrial fibrillation dose reductions may be required for the DOACs for stroke prevention
Extremes of body weight: Safety data is limited for DOACs in patients <50kg or >120kg
GFR <30: DOACs are metabolized renally so poor renal function can lead to toxic DOAC concentrations and bleeding.
Active cancer: For patients who are being considered for treatment for venous thromboembolism who have active cancer, DOACs should be avoided because they have not been studied rigorously in this patient population. LMWH remains the drug of choice for venous thromboembolism in patients with cancer.
Drug interactions: Anti-platelet agents, NSAIDs, phenytoin, carbemazepine, antifungals, marcolides, rifampin and anti-retrovirals in combination with DOACs may result in excess bleeding and/or upredictable drug levels.
Poor compliance: The half-life of DOACs are short (approximately 12 hours), and one missed dose may result in a normal coagulation system, increasing the risk for venous and arterial thrmobosis. There is no reliable way to monitor compliance as we do with INR monitoring in patients taking Warfarin.
Liver failure: Overt liver failure or liver enzymes >3 times the upper limit of normal are contraindications to DOACs.
Pregnancy: DOACs are contraindicated in pregnacy as they cross the placenta and may be teratogenic.
Any history of major bleeding
Baseline bloodwork before starting a DOAC in the ED
- CBC (to look for occult anemia, thrombocytopenia)
- Cr/GFR to screen for renal dysfunction as DOACs are partially metabolized through the kidney
- INR/PTT to screen for underlying bleeding or thrombotic disorder
- Liver panel for patients at risk for liver failure as DOACs are metabolized partially through the liver
How the DOACs work
Dabigatran works as a thrombin inhibitor inhibiting the conversion of fibrinogen to fibrin.
Rivaroxiban and Abixiban are Xa inhibitors and prevent the conversion of prothrombin to its active form thrombin.
Anticoagulation options for patient populations who have a contraindication to DOACs
For patients with active cancer or pregnancy: Full dose LMWH
For patients with severe renal failure: Full dose IV Heparin overlapping with Warfarin for at least 5 days AND until the INR is at least 2.0 for at least 2 days
For patients with body weight <50kg or >120kg: LMWH overlapping with Warfarin
Anticoagulation assays: Are there practical blood tests for detecting the presence or level of DOACS?
Are there any blood tests available in the ED that can help determine if a patient is taking their DOAC or determine whether or not their DOAC is still exerting anticoagulation effects in a bleeding patient?
While chromogenic Anti-Factor Xa does measure the concentration of the Xa Inhibitors (Rivaroxiban, Apixiban) it is not widely available in EDs. Dabigitran reliably elevates the dilute Thrombin Time (dTT), but it is not widely available in most EDs. However a normal Thrombin Time (TT), which most EDs do have access to, rules out Dabigitran anticoagulation effects.
The effects of DOACs on PT/INR or aPTT are variable. A normal PT/INR or aPTT does not rule out an anticoagulation effect.
The bottom line is that widely available anticoagulation assays are generally not useful clinically for measuring the anticoagulation effect of a DOAC.
DVT management with DOACs
DOACs have a fast onset within 1-2hrs so they are ideal initial drugs for patients who need to be anticoagulated immediately.
If there is a delay to definitive diagnosis via doppler compression ultrasound, patients with moderate to high suspicion of DVT (except those with a high risk of bleeding) should start anticoagulation therapy before the diagnosis is confirmed.
DOAC Dosing for venous thromboembolism
The first thing to know about DOAC dosing is that dosages are different for venous thromboembolism than for atrial fibrillation. Dosing for venous thromboembolism is fixed as opposed to atrial fibrillation where it is adjusted based on renal function, weight and age.
- Apixiban 10mg po bid for 1 week followed by 5mg po bid or
- Rivaroxiban 15mg po bid for 3 weeks followed by 20mg once daily
Isolated calf DVT: Do they require Anticoagulation?
Approximately 15-20% of identified isolated calf DVTs will progress proximally.
The suggested indications for anticoagulation treatment for isolated calf DVTs with anticoagulant are:
- All 3 deep veins involved
- Clot close to the popliteal vein
- Severe symptoms
- Clot > 5cm in length
- Previous venous thromboembolism
- Positive D-dimer
- Progression of the DVT on repeat imaging
Superficial Venous Thrombosis: Do they require Anticoagulation?
It’s first very important to understand that the Superficial Femoral Vein is not a superficial vein – it is a deep vein. So if you get an ultrasound doppler report that says there is a clot on the Superficial Femoral Vein, that needs to be treated as a DVT.
The suggested indications for anticoagulation treatment for superficial venous thrombosis are:
- Severe symptoms
- Clot close to sapheno-femoral junction
- >5cm in length
If you deem a patient with superficial venous thrombosis to be high risk for developing a DVT, low dose of LMWH (approx 1/3-2/5 the therapeutic dose) is the first line medication. DOACs can be considered as a second line option, however they have not been studied for this indication.
Anticoagulation with DOACs for patients with Atrial Fibrillation should start in the ED
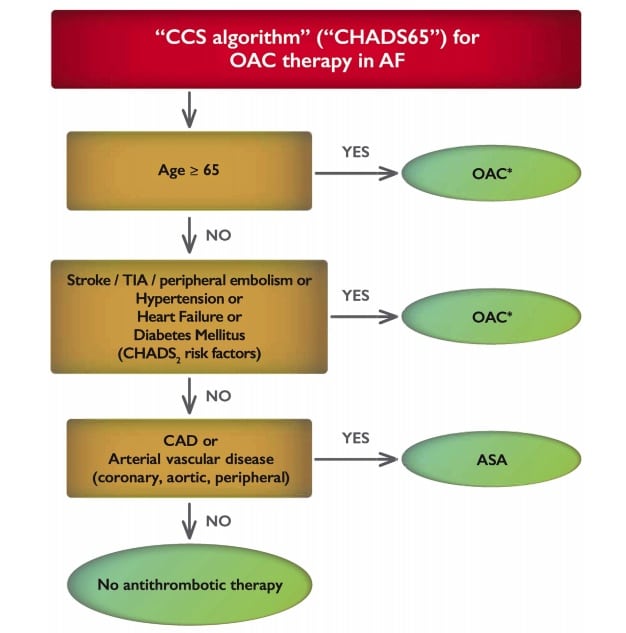
Oral anticoagulants for stroke prevention in patients with atrial fibrillation are currently recommended for:
All atrial fibrillation patients aged 65 or older or who have any one of the traditional CHADS2 risk factors of stroke, hypertension, heart failure, or diabetes (see algorithm below)
The Canadian Cardiovascular Society Guidelines on Atrial Fibrillation recommend that when oral anticoagulation therapy is indicated for patients with non-valvular atrial fibrillation, most patients should receive a DOAC in preference to warfarin.
DOACs are contraindicated in patients with valvular atrial fibrillation (mechanical valve or rheumatic valvular disease).
Ian Stiell on atrial fibrillation on EM Cases Episode 57 – The Stiell Sessions – Atrial Fibrillation
Bleeding risk of DOACs compared to Warfarin for stroke prevention in patients with atrial fibrillation
- The DOACs are at least as efficacious as Warfarin to prevent stroke in patients with atrial fibrillation
- The DOAcs are at least as safe as Warfarin in mitigating the risk of major bleeding
- The DOACs are associated with a lower risk of intracranial hemorrhage compared to Warfarin and when major bleeding occurs, DOACs are associated with a lower mortality rate
- The DOACs (in particular Dabigitran and Rivaroxiban) are associated with an increased risk of GI bleed compared to Warfarin
Risk-Benefit of DOACs in atrial fibrillation: Weighing CHADS2 score vs HAS-BLED score
The HAS-BLED Score for Major Bleeding Risk estimates the risk for major bleeding in patients on anticoagulation to help determine risk-benefit in atrial fibrillation care.
HAS-BLED Score
Hypertension History (uncontrolled, >160 mmHg systolic)
Renal Disease (Dialysis, transpant, Cr > 2.6 mg/dl or > 200 µmol/L)
Liver Disease (Cirrhosis, Bilirubin > 2x Normal, AST/ALT/AP > 3x Normal)
Stroke History
Prior Major Bleeding or Predisposition to Bleeding
Labile INR (Unstable/high INRs)
Age ≥ 65
Medication Usage Predisposing to Bleeding (Antiplatelet agents, NSAIDS)
Alcohol Usage History
Each factor scores 1 point with major bleeding risk of 3.4% with 1 factor and 9.1% for 5 factors.
Which patients taking antiplatelet agents should continue taking them when starting a DOAC?
The risk of major bleeding increases significantly in patients taking both antiplatelet agents and anticoagulants, and there is no added benefit of antiplatelet agents to prevent stroke in patient with Atrial Fibrillation.
For patients with a coronary stent placed within the last year on antiplatelet medications in whom you are considering stopping their antiplatelet agent and starting on an anticoagulant for stroke prevention in Atrial Fibrillation, our experts recommend consulting a cardiologist as there are many factors to consider such as whether the stent was a drug eluting stent, the length of the stent and risk of bleeding.
For patients with a coronary stent placed more than one year ago the Canadian Cardiovascular Society Guidelines recommend stopping the antiplatelet agent(s).
For patients taking Aspirin for primary prevention in whom an anticoagulant if being considered for stroke prevention, the Aspirin should be stopped as the risk of bleeding clearly outweighs the benefit of aspirin for primary prevention of ischemic events.
Dosing DOACs for stroke prevention in atrial fibrillation
Note that DOAC dosing for atrial fibrillation should be adjusted according to the patient’s age, weight and creatinine clearance as opposed to dosing for venous thromboembolism which is fixed.
Thrombosis Canada App for dosing DOACs in atrial fibrillation.
For adult patients with normal renal function and normal BMI the dosing for DOACs for stroke prevention in atrial fibrillation are:
- Dabigatran 150 mg twice daily, may be reduced to 110 mg twice daily if other risks for bleeding exist, or
- Rivaroxaban 20 mg once daily, or
- Apixaban 5 mg twice daily
Adjusting DOACs in patients with acute renal insufficiency
For patients with baseline normal renal function on a DOAC for stroke prevention with atrial fibrillation who become acutely ill with renal insufficiency, our experts recommend withholding one or two doses of DOAC until their renal function normalizes. Early follow-up to check their creatinine is important.
For patients with baseline normal renal function on a DOAC for a DVT or PE who become acutely ill with renal insufficiency, our experts recommend withholding their DOAC until their renal function normalizes, consider admitting them for monitoring and bridge them with LMWH.
In Episode 89 DOACs Part 2 – Bleeding and Reversal we will discuss the management of minor, moderate and major bleeding in patients taking DOACs as well as periprocedural adjustment of DOACs.
Dr. Himmel has no conflicts of interest to declare.
Dr. Douketis and Dr. Bell have received financial compensation for educational endeavours from companies that make DOACs.
Dr. Helman received financial compensation one time for participating in an educational video for the makers of Octaplex.
For more on DOACs on EM Cases:
Episode 36: Transfusions, Anticoagulants and Bleeding
Episode 37: Anticoagulants, PCCs and Platelets
Episode 89 – DOACs Part 2: Bleeding and Reversal Agents
Best Case Ever 18: Anticoagulant Reversal in Trauma
Best Case Ever 51 – Anticoagulants and GI Bleed with Walter Himmel
References
Raja AS, Geyer B. Emergency department management of patients on novel oral anticoagulant agents. Emerg Med Pract. 2013;15(10):1-18.
Ruff CT, et al. Comparison of the efficacy and safety of new oral anticoagulants with warfarin in patients with atrial fibrillation: a meta-analysis of randomised trials. Lancet 2014;383(9921):955-962.
Baglin T. The role of the laboratory in treatment with new oral anticoagulants. J Thromb Haemost 2013;11(Suppl 1):122-128.
Cuker A et al. Laboratory measurement of the anticoagulant activity of the non-vitamin K oral anticoagulants. J Am Coll Cardiol 2014;64(11):1128-1139.
Schwarz T, Buschmann L, Beyer J, Halbritter K, Rastan A, Schellong S. Therapy of isolated calf muscle vein thrombosis: a randomized, controlled study. J Vasc Surg. 2010;52(5):1246-50.
Kearon C, et al. Antithrombotic therapy for VTE disease: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest 2012;141(2 Suppl):e419S-e494S.
van der Hulle T, et al. Effectiveness and safety of novel oral anticoagulants as compared with vitamin K antagonists in the treatment of acute symptomatic venous thromboembolism: a systematic review and meta-analysis. J Thromb Haemost 2014;12(3):320-328.
Utter GH, Dhillon TS, Salcedo ES, et al. Therapeutic Anticoagulation for Isolated Calf Deep Vein Thrombosis. JAMA Surg. 2016;151(9):e161770.
Horner D, Hogg K, Body R, Nash MJ, Baglin T, Mackway-jones K. The anticoagulation of calf thrombosis (ACT) project: results from the randomized controlled external pilot trial. Chest. 2014;146(6):1468-77.
Graham DJ, Reichman ME, Wernecke M, et al. Stroke, Bleeding, and Mortality Risks in Elderly Medicare Beneficiaries Treated With Dabigatran or Rivaroxaban for Nonvalvular Atrial Fibrillation. JAMA Intern Med. 2016.
Macle L, Cairns J, Leblanc K, et al. 2016 Focused Update of the Canadian Cardiovascular Society Guidelines for the Management of Atrial Fibrillation. Can J Cardiol. 2016;32(10):1170-1185. Full PDF
Lip GY, Frison L, Halperin JL, Lane DA. Comparative validation of a novel risk score for predicting bleeding risk in anticoagulated patients with atrial fibrillation: the HAS-BLED (Hypertension, Abnormal Renal/Liver Function, Stroke, Bleeding History or Predisposition, Labile INR, Elderly, Drugs/Alcohol Concomitantly) score. J Am Coll Cardiol. 2011;57(2):173-80.
Airaksinen KE, Grönberg T, Nuotio I, et al. Thromboembolic complications after cardioversion of acute atrial fibrillation: the FinCV (Finnish CardioVersion) study. J Am Coll Cardiol. 2013;62(13):1187-92.
Other FOAMed Resources for DOACs Use and Misuse
Thrombosis Canada clinical guidelines app
Intensive Care Network podcast Bloody Anticoagulants
Get the NOAC knowhow on St. Emlyn’s blog
Dr. Himmel’s Best Case Ever on GI Bleed with Dabigitran
EM Cases Episode 37 – Anticoagulants, PCCs and Platelets
Ian Stiell on atrial fibrillation on EM Cases Episode 57 – The Stiell Sessions – Atrial Fibrillation
Now test your knowledge with a quiz.




Fabulous podcast! so relevant and easy to follow. thank you! I’m an SRMO (senior resident) in Emergency in Australia.